Selfish X-linked alleles and X chromosome inactivation
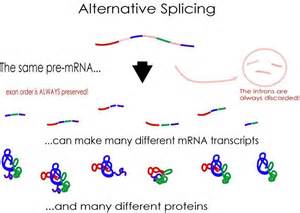
(click to enlarge)
From Fertilization to Adult Sexual Behavior
In our section on molecular epigenetics from 1996 we wrote:
Genomic-imprinting is also manifest in specific parts of the X-inactivation region’s related XIST gene. Here male- and female-specific methyl-group patterns participate in X-inactivation in females and also in the preferential inactivation of the paternal X in human placentae of female concepti (Harrison, 1989; Monk, 1995). This process indicates that tissues of the early conceptus can sense and react differentially to epigenetic sexual dimorphisms on the female conceptus’ own two X chromosomes. Furthermore, variations of X-inactivation patterns often account for traits discordance in monozygotic twin females. In other words, they are often found to have nonidentical patterns of X-inactivation, yielding differing expression of noticeable X-linked traits (Machin, 1996).
The pseudoscientific nonsense about selfish genes has prevailed despite proof of how the food energy-dependent pheromone-controlled physiology of reproduction biophysically constrains viral latency in the context of RNA-mediated amino acid substitutions and cell type differentiation in all cell types of all individuals of all living genera.
The focus of pseudoscientists is still on population genetics and mathematical models that have no explanatory power. Finally, I understand why Roger Gorski retired. Twenty-seven years ago, he wrote:
While the hypothalamic LRF pulse generatior pituitary gonadotropin gondal axis has a fundamental commonality to all mammalian species, the specialization that has evolved in different species merits emphasis.” “While photoperiodic cues and pheromones play an important role in some species, they do not in the human being.” “It is possible that the LRF neurosecretory neuron network itself is the pacemaker. – p 281. Maturation of neural mechanisms and the pubertal process. Control of the Onset of Puberty. Grumback, M.M., Sizonenko, P.C., Aubert, M.L 1990

I met Roger Gorski in 1995 and I discussed at length with him the publication of The Scent of Eros: Mysteries of Odor in Human Sexuality. (1995/2002) The link to pheromone-controlled sexual differentiation of cell types in species from yeasts to primates became perfectly clear.
By then, I think he knew how difficult it would be to teach others the facts after contradicting himself. It made no sense for “the godfather of neuroscience” to try. For the most recent confirmation of that fact, see:
Intragenomic conflict and immune tolerance: do selfish X-linked alleles drive skewed X chromosome inactivation? (2017)
…studying immunological and reproductive differences in female mice with genetically-‐driven SXCI could answer important questions as to the broader phenotypic consequences of SXCI. Population genomic studies of regions implicated in SXCI may help us to understand the evolutionary forces driving SXCI-‐favoring alleles.
Intragenomic conflict and cancer (2002)
Intragenomic conflict occurs when some elements within the genome produce effects that enhance their own probability of replication or transmission at the expense of other elements within the same genome. Here it is proposed that mutations involved in intragenomic conflict are particularly likely to be co-opted by evolving lineages of cancer cells, and hence should be associated with the occurrence of cancer. We discuss several types of intragenomic conflict that are associated with various forms of cancer.
During the past 15 years, “intragenomic conflict” has been placed into the context of energy-dependent differences in the microRNA/messenger RNA that all serious scientists have linked from changes in electrons to ecosystems via base editing and RNA editing. Only pseudoscientists have failed to make any progress whatsoever, and they all have been stuck with their ridiculous theories.
Want more on the same topic?
Swipe/Drag Left and Right To Browse Related Posts:
NGS vs 5th generation warfare (4)
< 1 MIN READ
0
miRNA-mediated predictions (5)
3 MIN READ
0
miRNA-mediated predictions (4)
2 MIN READ
0
Minimal level of conflict (8)
4 MIN READ
0
Total recall 6000 years (7)
2 MIN READ
0
Biogeochemical is geopolitical (8)
2 MIN READ
0
Biogeochemical is geopolitical (4)
3 MIN READ
0
WHO broke the breakthrough (2)
2 MIN READ
0
miRNA-mediated epigenetic effects (7)
< 1 MIN READ
0
The devil is in the dirt (2)
2 MIN READ
0
The devil is in the dirt (1)
2 MIN READ
0
Scientism, atheism and theology (10)
2 MIN READ
0
Epistemic corruption (8)
2 MIN READ
0
The microRNA-mediated future of humanity (3)
3 MIN READ
0
CDC’s ignorance: from roots to shoots (3)
4 MIN READ
0
microRNA-mediated sex differences (1)
2 MIN READ
0
MicroRNA-mediated hypoxia vs cancer (1)
5 MIN READ
0
Weaponized health communication (3)
4 MIN READ
0
Genetic endemism: apologetics vs epigenetics (1)
2 MIN READ
0
Quantum initiation of cold chemistry vs Hypeology (3)
3 MIN READ
0
The eternal significance of microRNAs and the Vietnam Memorial (1)
3 MIN READ
0
Epigenetic inheritance of spatiotemporal regulation
4 MIN READ
0
The eternal significance of microRNAs (7)
3 MIN READ
0
The eternal significance of microRNAs (5)
2 MIN READ
0
Ecological adaptations vs the randomness of evolution (3)
4 MIN READ
0
Environmental selection is natural selection (2)
2 MIN READ
0
2018 March for Science vs microRNAs
7 MIN READ
0
The eternal significance of microRNAs (1)
< 1 MIN READ
0
Sympatric Speciation vs pseudosceintific nonsense (4)
2 MIN READ
0
Sympatric Speciation vs Pseudoscientific Nonsense (1)
4 MIN READ
0
Complexity: Routes and Patterns (4)
< 1 MIN READ
0
Laws of Biology / Rules of Life
3 MIN READ
0
Part 3: Light-controlled cell biology (revisited)
3 MIN READ
0
Ecological adaptation: a new definition of heredity (4)
4 MIN READ
0
Conceptual critique: Innateness vs the death gene (1)
5 MIN READ
0
Cryo-EM: Linking spatial and conformational constraints
9 MIN READ
0
BiondVax Universal Flu Vaccine Patent
4 MIN READ
0
The MicroRNAome Strikes Back: A Sokalian hoax (4)
5 MIN READ
0
The MicroRNAome Strikes Back: A Sokalian hoax (2)
6 MIN READ
0
A mental health problem at the highest level (2)
3 MIN READ
0
Diet-driven RNA interference and cancer prevention (3)
6 MIN READ
0
Enzyme-constrained interethnic diversity (8)
< 1 MIN READ
0
Enzyme-constrained interethnic diversity (7)
6 MIN READ
0
Diet-driven RNA interference and cancer prevention
5 MIN READ
0
Quantized energy-dependent viral trophism (2)
2 MIN READ
0
Who created your virus-driven death gene? (3)
3 MIN READ
0
Kinetically Stable Thermodynamically Activated Cell Metabolism (3)
3 MIN READ
0
The tipping point (revisited): 68,000 publications
5 MIN READ
0
Narcissistic egomaniacal and deadly denial of autophagy
6 MIN READ
0
Elsevier fails to support the concept of autophagy
4 MIN READ
0
From Autophagy.pro (transitions)
4 MIN READ
0
Nature vs Science and Autophagy.pro
7 MIN READ
0
The overwhelming ignorance of sex researchers (2)
4 MIN READ
0
Energy-dependent base editing and correction of mutations (2)
9 MIN READ
0
Trashing the 2nd Law
7 MIN READ
0
The overwhelming ignorance of sex researchers
5 MIN READ
0
Who created your virus-driven death gene? (1)
5 MIN READ
0
Eutrophication and phosphorylated biodiversity
6 MIN READ
0
Alternative splicing: a new therapy?
3 MIN READ
0
Pheromones biophysically constrain base editing and RNA editing
3 MIN READ
0
From base editing to RNA editing (5)
< 1 MIN READ
0
From base editing to RNA editing (4)
5 MIN READ
0
The next reformation: 500 years too late? (3)
5 MIN READ
0
Cryo-EM: More than a suggestion
< 1 MIN READ
0
Exome Sequencing Impact in Routine Care
4 MIN READ
0
Virulence: Out of stock
2 MIN READ
0
Predicting who wins the 2017 Nobel Prizes (2)
4 MIN READ
0
Predicting who wins the 2017 Nobel Prizes
4 MIN READ
0
Sexual communication signals: New Insights!
10 MIN READ
0
Stem cells do not create themselves
3 MIN READ
0
Pseudoscientists hate what science explains! (3)
4 MIN READ
0
Your indifference is killing you and others (4)
4 MIN READ
0
Robert Sapolsky’s legacy of atheistic pseudoscientific nonsense
4 MIN READ
0
Energy-dependent physical and biophysical constraints (9)
3 MIN READ
0
Energy-dependent physical and biophysical constraints (8)
5 MIN READ
0
Energy-dependent physical and biophysical constraints (5)
7 MIN READ
0
Energy-dependent physical and biophysical constraints (3)
2 MIN READ
0
Energy-dependent physical and biophysical constraints (2)
4 MIN READ
0
Hard Science vs the neo-Darwinian “magic box”
9 MIN READ
0
Food energy-dependent epigenetic adaptation
11 MIN READ
0
Energy-dependent microRNA biogenesis
4 MIN READ
0
Open Science: Closed to facts about microRNAs
6 MIN READ
0
Viruses in pathogenic variants disrupt alternative splicings (2)
3 MIN READ
0
Epigenetically-effected nucleosome repositioning sheds Dobzhansky’s light on evolution
3 MIN READ
0
Cytosis: Biology Content
7 MIN READ
0
Light-activated feedback loops vs self-organization of ecosystems
7 MIN READ
0
Proof: Sunlight is energy as information
< 1 MIN READ
0
The essence of precision medicine
4 MIN READ
0
Allen Institute confirms food energy is information
6 MIN READ
0
Theistic evolutionists fight back and lose (3)
4 MIN READ
0
Charles Darwin refutes theistic evolution
2 MIN READ
0
Pseudoscientists fail to refute theistic evolution
4 MIN READ
0
Francis S. Collins refutes theistic evolution
6 MIN READ
0
Combating evolution: Battlefield medicine vs politicized science (2)
3 MIN READ
0
Physicists: Desperate Acts (revisited)
6 MIN READ
0
Dobzhansky 1973 and Precision Medicine (2)
5 MIN READ
0
Twisted theories and weaponized facts
3 MIN READ
0
Autophagy: from pre-mRNAs to microRNAs, enhancers, QTLs et al.
6 MIN READ
0
Science journalists or paid propagandists? (3)
6 MIN READ
0
Energy-dependent alternative splicings 1996 – 2016 (2)
5 MIN READ
0
2016 obfuscated facts about energy as information
5 MIN READ
0
Re-inventing mutation-driven evolution
3 MIN READ
0
Bird-brained theorists
< 1 MIN READ
0
Autophagy is the antiphage defense strategy
3 MIN READ
0
Energy-dependent de novo creation and neurogenesis
7 MIN READ
0
Theories vs facts about polycombic adaptation
3 MIN READ
0
The futility of The Battlefield FB group
10 MIN READ
0
Controlled amino acid treatment of all pathology
4 MIN READ
0
Survivors of RNA-mediated terrorism
5 MIN READ
0
RNA-mediated “repurposing” is autophagy
2 MIN READ
0
RNA-mediated "repurposing" is autophagy
2 MIN READ
0
Epigenetics and autophagy vs mutations and evolution (8)
4 MIN READ
0
Energy-dependent maternal-to-zygotic transition
6 MIN READ
1
Virus-driven mutation or amino acid substitution
5 MIN READ
0
Epigenetics and autophagy vs mutations and evolution (7)
3 MIN READ
0
Pheromone-controlled autophagy
4 MIN READ
0
Virus-mediated hecatombic evolution
2 MIN READ
0
Nutrient-dependent autophagy
3 MIN READ
0
The last RNA-mediated theory killer
< 1 MIN READ
0
Hydrogen-atom energy in DNA base pairs
6 MIN READ
0
Light energy-dependent active motifs
6 MIN READ
0
Light energy-induced base pair changes (2)
4 MIN READ
0
Light energy-induced base pair changes (1)
6 MIN READ
0
How did the innate immune system evolve?
4 MIN READ
0
Increased soil pH and nutrient availability
4 MIN READ
0
Energy-dependent cellular communication
3 MIN READ
0
Antithetical conclusions (7)
3 MIN READ
0
Non-random pheromone-controlled cell type differentiation
3 MIN READ
0
Energy-dependent natural fluorescence and bioluminescence
2 MIN READ
0
Biophotonics, glycobiology, quantized biodiversity (3)
< 1 MIN READ
0
Biophysically constrained cell type differentiation
3 MIN READ
0
From angstroms to ecosystems
7 MIN READ
0
Food supplement or licensed immunostimulant?
6 MIN READ
0
Q and A: Energy-dependent cell type differentiation
10 MIN READ
0
Virus-driven downsizing of the human brain
2 MIN READ
0
Major transition ends use of silly theories
5 MIN READ
0
RNA methylation
2 MIN READ
0
RNA-mediated physics, chemistry, and molecular epigenetics (5)
6 MIN READ
0
RNA-mediated physics, chemistry, and molecular epigenetics (4)
5 MIN READ
0
RNA-mediated physics, chemistry, and molecular epigenetics (3)
5 MIN READ
0
Wasted Templeton Funding (3)
2 MIN READ
0
Millions of water molecules vs years of evolution
2 MIN READ
0
Ricki Lewis’ Time Machine (4)
8 MIN READ
0
Selective reporting of inferences: examples of pseudoscience
14 MIN READ
0
Energy dependent RNA-mediated immunity (5)
4 MIN READ
0
Energy-dependent RNA-mediated immunity (1)
10 MIN READ
0
Science vs semantics
4 MIN READ
0
Soil bacteria, bulls, cows, microRNAs, and mammary glands
3 MIN READ
0
Hybrids: making microbes and Democrats into monkey’s uncles?
11 MIN READ
0
Creating gravity, nucleic acids, receptors, and supercoiled DNA (2)
9 MIN READ
0
Cancer: Evolution 2.0’s Blind Spot
13 MIN READ
0
Ricki Lewis’ Time Machine
5 MIN READ
0
Stress-induced energy-dependent DNA repair
4 MIN READ
0
A failed theory of cancer: two more decades of pseudoscience
5 MIN READ
0
Ecological genomics: teleophobes respond (too late)
10 MIN READ
0
Natural cooperation and Evolution 2.0
6 MIN READ
0
MicroRNAs vs Red Queen hypothesis
4 MIN READ
0
Theorists can’t understand biology
6 MIN READ
0
Receptor methylation controls behavior
2 MIN READ
0
MicroRNA-encoded behavior
7 MIN READ
0
The virome, microbiome, replisome and supercoiled DNA
2 MIN READ
0
Skip the politics; embrace the facts
5 MIN READ
0
Mystery machine vs medical intelligence
2 MIN READ
0
Xist-ing on planet Earth
2 MIN READ
0
Let there be anti-entropic light (2)
4 MIN READ
0
Is mainstream science in “Science” pseudoscience?
5 MIN READ
0
Mechanisms of stress: from genes to cancer
9 MIN READ
0
Theorists have not seen the light
4 MIN READ
0
A 5-10K comparison of design principles to evolution
3 MIN READ
0
Alternative pre-mRNA splicing and ecological adaptation
7 MIN READ
0
“New” quantum biology. Pirating the old
3 MIN READ
0
Anti-entropic containment of energy: symbiosis 1.0
5 MIN READ
0
RNA-mediated terms of virus-induced en-deer-ment
2 MIN READ
0
Hematopoiesis and practopoiesis
4 MIN READ
0
Becoming biologically informed (3)
3 MIN READ
0
Easy editing: Reinventing our RNA world
3 MIN READ
0
“New” epigenetic mechanism for lifelong learning?
3 MIN READ
0
Protein folding and Google page rank
2 MIN READ
0
Living the life that randomness created? (Sarcasm alert)
3 MIN READ
0
Epigenetic regulation of aging by glycine and GnRH
5 MIN READ
0
Pattern recognition: biogeochemical structure and function
5 MIN READ
0
Informing the biologically uninformed
< 1 MIN READ
0
Batch effect vs epigenetic effects
3 MIN READ
0
Bee-birthed epigenetics and primate cell types
5 MIN READ
0
Virus-driven cancer treatment and prevention
2 MIN READ
0
A special issue on nutritional epigenetics
5 MIN READ
0
Retinoic acid + one receptor regulate the genome
3 MIN READ
0
From gut bacteria to breast milk and back
5 MIN READ
0
Epigenetics vs the fossil record
4 MIN READ
0
Re-inventing a completely new thing
4 MIN READ
0
RNA-mediated cell types and precision medicine
5 MIN READ
0
Misunderstanding cancer
7 MIN READ
0
Nutrient-dependent microRNAs control cell types
6 MIN READ
0
2 genes in 2 species (too expensive and too insignificant)
11 MIN READ
0
Quantum correlations/pseudoscience
3 MIN READ
0
Anti-entropic solar energy
5 MIN READ
0
Correctly modeling biological energy
2 MIN READ
0
RNA-directed gene choice
5 MIN READ
0
What about birds?
5 MIN READ
0
Reverse phosphorylation
7 MIN READ
0
RNA-mediated repurposing in microbes and adaptations in primate brains
2 MIN READ
0
RNA-mediated “repurposing” is nutrient-dependent and pheromone-controlled
3 MIN READ
0
RNA-mediated "repurposing" is nutrient-dependent and pheromone-controlled
3 MIN READ
0
Let there be anti-entropic light (1)
11 MIN READ
0
An epigenetic trap (the prequel)
5 MIN READ
0
Imagining that data historically supports evolutionary theory
7 MIN READ
0
The anti-entropic force of "Nature"
2 MIN READ
0
Nutritional epigenetics, exercise, and immune system integrity
4 MIN READ
0
Atoms to ecosystems is not almost a molecular ecology
3 MIN READ
0
Epigenetic effects of viruses on cellular homeostasis (2)
2 MIN READ
0
Biological energy and a microbiome model of a light-driven time machine
3 MIN READ
0
Quantum Superpositions: let there be light
4 MIN READ
0
Are viruses microRNAs? (2)
5 MIN READ
0
Sneaking up from behind
4 MIN READ
0
Beneficial microbes kill beneficial mutations
4 MIN READ
0
Constrained evolution is ecological adaptation
5 MIN READ
0
A single amino acid substitution differentiates cell types of E. coli
3 MIN READ
0
Unconstrained evolutionary innovability
4 MIN READ
0
Mutagenesis: Replacing facts with theories
5 MIN READ
0
ISHE's human ethology group
2 MIN READ
0
From Hydra to humans vs a Lakatosian research program
4 MIN READ
0
Unified nutritional and molecular mechanisms
9 MIN READ
0
Model organisms: the birds and the bees
3 MIN READ
0
Chemical ecology and RNA-mediated control of DNA loops
< 1 MIN READ
0
Jumping back: Science or Pseudoscience?
6 MIN READ
0
Single-cell level assay of protein biosynthesis and degradation
2 MIN READ
0
Dual genomes: exposing the evolution industry
5 MIN READ
0
RNA-mediated species specificity
2 MIN READ
0
Are mutations beneficial?
4 MIN READ
0
Ecological adaptations reported as evolution in insects and mammals
4 MIN READ
0
Biologists puzzled by evolved RNAs and decaying DNA
6 MIN READ
0
Nothing new under the sun, except pseudoscientific nonsense
6 MIN READ
0
Making sense of quotes scattered across disciplines
2 MIN READ
0
Intelligent viruses and cancers?
10 MIN READ
0
Sexual differentiation of cell types in plants
2 MIN READ
0
A model of MHC 'evolution'
2 MIN READ
0
From deep time into real time: What evolutionary processes?
10 MIN READ
0
Are evolutionary theorists 'nob ends'?
5 MIN READ
2
No excuses: Creation and the meaning of organismal complexity
7 MIN READ
0
Nutrient-dependent gene duplication in plants (but not animals?)
4 MIN READ
0
Behavioral ecology: please continue to believe in our fantasies
3 MIN READ
0
RNA-mediated genetic engineering (Part 2)
3 MIN READ
0
RNA-mediated genetic engineering
2 MIN READ
0
Color vision refutes the evolutionary dogma of gene duplication
4 MIN READ
0
Forces of "Nature" limit dissemination of information
4 MIN READ
0
Genomic surveillance ends our world of RNA-mediated ecological adaptations
2 MIN READ
0
Metabolism, fixation, health or neurodegerative disorder
2 MIN READ
0
Physics, Chemistry, and Molecular biology (PCMb)
2 MIN READ
0
Ecological variation and niche construction: 1, 2, 3
6 MIN READ
0
Stop evolutionary theorists. Kill cancers
3 MIN READ
0
De novo gene Creation sans evolution of genes via mutations
4 MIN READ
0
Did our adapted mind evolve?
6 MIN READ
0
Seemingly futile cycles are not thermodynamically futile
3 MIN READ
0
Memory of repression and memory of behavior
2 MIN READ
0
Exploding genomes and chromosomal rearrangements via RNA-mediated events
2 MIN READ
0
Behavior (2): All responses are RNA-mediated not genetically-determined
4 MIN READ
0
Baby talk: More misrepresentations of ecological adaptations
2 MIN READ
0
Epimutations: Attacking pseudoscientific dogmas
4 MIN READ
0
Order and disorder: Ecological adaptations not mutations
6 MIN READ
0
The quantum biology of consciousness
3 MIN READ
0
Cell-type differentiation
2 MIN READ
0
96 fixed amino acid substitutions, not 96 genes
6 MIN READ
0
Social experiences epigenetically effect gene networks
2 MIN READ
0
Pheromone-controlled thermodynamics and cancer
2 MIN READ
0
Honeybees, food odors, and perfume
< 1 MIN READ
0
Human Pheromones
< 1 MIN READ
0
Study Results: Human pheromones increase flirtatious behavior and ratings of attraction
2 MIN READ
0
Human pheromones work by effecting hormones that affect behavior
2 MIN READ
0
Conditioned sexual arousal to odor
2 MIN READ
0
Scent of Eros pheromone-enhanced fragrance products
2 MIN READ
0
Welcome to Pheromones.com
< 1 MIN READ
0



[…] For an update, see: Selfish X-linked alleles and X chromosome inactivation11/6/17 […]