Epigenetic regulation of aging by glycine and GnRH
(click to enlarge)
Summary: “…the regulation of two genes involved with the production of glycine, the smallest and simplest amino acid, is partly responsible for some of the characteristics of aging. This indicates that the aging process in the mitochondrion is controlled by epigenetic regulation, not by mutations.”
My comment: The broad-based extension of the fact that aging is epigenetically controlled, extends everything known about RNA-mediated cell type differentiation across the life history transitions of all genera. The focus here is on vertebrates, but the conserved molecular mechanisms extend across all species.
———————————
Thanks to Teresa Binstock for alerting me to this. Epigenetic regulation of the nuclear-coded GCAT and SHMT2 genes confers human age-associated mitochondrial respiration defects
Excerpt:
Given that human aging can be seen as a consequence of a programmed phenomenon, it is possible that epigenetic regulation also controls human aging.
My comment: See: Search Results for ‘glycine’ here at RNA-mediated.com and also Search Results for: glycine in the “Science” section at Pheromones.com for additional information. It should become apparent why I started with the domain Pheromones.com in 1995 and linked nutrient-dependent RNA-mediated amino acid substitutions to cell type differentiation after addressing the control of nutrient-dependent reproduction. Others now realize that the physiology of reproduction biophysically constraints transgenerational epigenetic inheritance of morphological and behavioral phenotypes. The new information about the control of RNA-mediated cell type differentiation by pheromones and anti-aging medicine has been delayed for more than two decades by human pheromone-deniers and other pseudoscientists.
In 1994, for example: I presented “Olfactory-hormonal relationships in learning, memory, aging, and behavior” during the “2nd Annual Conference on Anti-aging Medicine & Biomedical Technology for the year 2010” and in 1995, I presented “Olfactory-genetic-neuronal-hormonal reciprocity in learning, memory, behavior and in immune function” at the “3rd Annual Conference on Anti-aging Medicine & Biomedical Technology for the year 2010.”
Teresa Binstock’s prescient contributions on RNA-mediated cell type differentiation in our 1996 Hormones and Behavior review article led me to examine the role of achiral glycine in vertebrates. See: From Fertilization to Adult Sexual Behavior
Excerpt:
Evolutionary conservation, both of pheromonal communication and its importance to behavior, is indicated by the involvement of a key mammalian reproductive hormone. For instance, a yeast pheromone, the alpha-mating factor, is very similar in structure to mammalian gonadotropic releasing hormone (GnRH). When injected into rats, this chemical binds to pituitary GnRH receptors and brings about the release of LH. Loumaye, Thorner, and Catt (1982) note: “GnRH and the yeast alpha-mating factor appear to represent a highly conserved effector system which includes the peptide ligand, the cell-surface receptor, and the physiological regulation of reproductive function” (p. 1325).
My comment: Substitution of the only achiral amino acid in the GnRH decapeptide of vertebrates links the light-induced de novo creation of glycine and other amino acids to the nutrient-dependent pheromone-controlled behaviors of species from microbes to humans. One need only consider that fact in the context of what is currently known about the biophysically constrained chemistry of RNA-mediated amino acid substitutions and protein folding in all genera.
1994 Abstract
The early prenatal migration of gonadotropin releasing hormone (GnRH) neurosecretory neurons appears to enable a neuroendocrine sequence of events that allows human pheromones to influence postnatal GnRH secretion, maturation of the hypothalamic-pituitary-gonadal axis; and, in part, the hypothalamic-pituitary-adrenal axis; hormone-dependent synaptogenesis and synaptolysis; neurotransmission; learning; memory; and behavior. That GnRH regulates the collective neural output manifest in reproductive behavior seems consistent with effects of drug therapies that influence the GnRH pulse, and which are used to treat disorders of neuroendocrine and reproductive maturation as well as dysfunctional behaviors. Is the hypothalamic GnRH pulse generator both the biologic and the psychologic core of mammalian reproduction? What is the contribution of extrahypothalamic GnRH? Is there a lack of “hard” scientific evidence for relationships between biologically relevant odors, olfaction, aging, and human behavior?
1995 Abstract
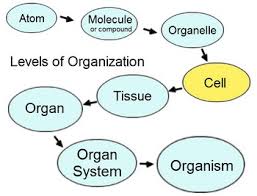
A five-step pathway allowing the social environment (“nurture”) to influence the genetic substrates (“nature”) of mammalian behavior is: gene->cell->tissue->organ->organ system. Though there are many environmental influences on the first step of this pathway, odors are the only known social-environmental stimuli that appear to activate gene expression in neurosecretory cells of tissue in the brain an organ that is essential to any organ system involved in learning, memory, and behavior. Olfaction appears to influence learning, memory, and behavior. Thus, the production and distribution of human odors may link two aspects of our social environment (e.g., olfaction and odors) to the genetic substrates of our behavior through a five-step pathway common to many other vertebrates. Olfactory input influences the gonadotropin-releasing hormone (GnRH)-directed regulation of gonadal and adrenal steroidogenesis. Thus, olfactory deficits associated with aging may be linked to a need for hormone replacement therapy, including dehydroepiandrosterone (DHEA). Similarly, olfactory deficits may be linked to immune system function. Many other hormones/neurotransmitters (e.g., melatonin and dopamine) feed back on the GnRH neuronal pathway. This pathway appears to be both the biological and the psychological core of mammalian, including human, behavior. Thus, the influence of odors and olfaction on levels of hormones, including neurotransmitters, may be linked to age-related changes in learning, memory, behavior, and immune system function.
2015
Excerpts:
…the regulation of two genes involved with the production of glycine, the smallest and simplest amino acid, is partly responsible for some of the characteristics of aging.This indicates that the aging process in the mitochondrion is controlled by epigenetic regulation, not by mutations.
See for comparison: Mutation-Driven Evolution
Concluding sentences:
“In other words, genomic conservation and constraint-breaking mutation is the ultimate source of all biological innovations and the enormous amount of biodiversity in this world. In this view of evolution there is no need of considering teleological elements” (p. 199).
See for comparison: Nutrient-dependent/pheromone-controlled adaptive evolution: a model
Concluding paragraph:
Unconscious affects that are manifested during the development of diversified life and human behavior are, by their very nature, part of life that few people think about (Kohl et al., 2001). Therefore, the largest contributor to the development of our personal preferences may be the unconscious epigenetic effects of food odors and pheromones on hormones that organize and activate behavior. If so, the model represented here is consistent with what is known about the epigenetic effects of ecologically important nutrients and pheromones on the adaptively evolved behavior of species from microbes to man. Minimally, this model can be compared to any other factual representations of epigenesis and epistasis for determination of the best scientific ‘fit’.
My comment: All accurate representations of biologically-based cause and effect have consistently shown that there are no other factual representations of epigenesis and epistasis that can be compared to my model. The most recent report on glycine and cell type differentiation during the life history transitions of vertebrates also suggests that the honeybee model organism links nutrient-dependent base pair substitutions in yeasts to the RNA-mediated amino acid substitutions in vertebrates that differentiate nutrient-dependent pheromone-controlled cell types via the conserved molecular mechanism that Teresa Binstock detailed in the “molecular epigenetics” section of From Fertilization to Adult Sexual Behavior.
Excerpt:
Disease is associated with mutations exemplified in cancer where perturbations of the glucose-dependent thermodynamic/thermoregulatory equilibrium are equally clear (Locasale,
2012).
Excerpt:
Reprogramming of gene expression in elderly fibroblasts occurred in GCAT (Fig. 3b), which regulates glycine production in mitochondria17, 18. It was therefore likely that reduced glycine production in mitochondria by epigenetic downregulation of GCAT (Fig. 3a) resulted in the age-associated respiration defects (Fig. 1a).
My comment: Reference 17, is Locasale, J. W. Serine, glycine and one-carbon units: cancer metabolism in full circle. Nat. Rev. Cancer 13, 572–583; doi:10.1038/nrc3557 (2013).
The barrage of pseudoscientific nonsense touted by evolutionary theorists and their idiot minions continues to test the patience of anyone who challenges ideas about cell type differentiation that link mutations to pathology and to aging, and RNA-mediated amino acid substitutions to health and longevity in all genera.
See for examples of nonsense touted by the biologically uninformed:
In evolution, ‘house of cards’ model wins
Tiny spheres of human cells mimic the brain, researchers say
Our bond with dogs may go back more than 27,000 years
John Glenn: Evolution should be taught in schools
Want more on the same topic?
Swipe/Drag Left and Right To Browse Related Posts:
Energy-as-information & the aurora borealis (2)
2 MIN READ
0
Energy-as-information & the aurora borealis (1)
< 1 MIN READ
0
Pea protein, p53 and cancer prevention (10)
3 MIN READ
0
Pea protein, p53 and cancer prevention (9)
3 MIN READ
0
Pea protein, p53 and cancer prevention (7)
2 MIN READ
0
Pea protein, p53 and cancer prevention (5)
2 MIN READ
0
Pea protein, p53 and cancer prevention (4)
< 1 MIN READ
0
Pea protein, p53 and cancer prevention (3)
2 MIN READ
0
Pea protein, p53 and cancer prevention (2)
2 MIN READ
0
Pea protein, p53 and cancer prevention (1)
2 MIN READ
0
The bioweapons cartel (10)
< 1 MIN READ
0
The bioweapons cartel (8)
2 MIN READ
0
The bioweapons cartel (7)
< 1 MIN READ
0
The bioweapons cartel (6)
2 MIN READ
0
The bioweapons cartel (5)
5 MIN READ
0
The bioweapons cartel (4)
2 MIN READ
0
The bioweapons cartel (3)
4 MIN READ
0
The bioweapons cartel (2)
2 MIN READ
0
The bioweapons cartel (1)
4 MIN READ
0
Whistleblowers found: dead or alive (10)
2 MIN READ
0
Whistleblowers found: dead or alive (9)
2 MIN READ
0
Whistleblowers found: dead or alive (8)
2 MIN READ
0
Whistleblowers found: dead or alive (7)
< 1 MIN READ
0
Whistleblowers found: dead or alive (6)
2 MIN READ
0
Whistleblowers found: dead or alive (5)
2 MIN READ
0
Whistleblowers found: dead or alive (4)
< 1 MIN READ
0
Whistleblowers found: dead or alive (3)
2 MIN READ
0
Whistleblowers found: dead or alive (2)
3 MIN READ
0
Whistleblowers found: dead or alive (1)
3 MIN READ
0
NGS vs 5th generation warfare (10)
5 MIN READ
0
NGS vs 5th generation warfare (9)
2 MIN READ
0
NGS vs 5th generation warfare (8)
< 1 MIN READ
0
NGS vs 5th generation warfare (7)
3 MIN READ
0
NGS vs 5th generation warfare (6)
2 MIN READ
0
NGS vs 5th generation warfare (5)
3 MIN READ
0
NGS vs 5th generation warfare (4)
< 1 MIN READ
0
NGS vs 5th generation warfare (3)
< 1 MIN READ
0
NGS vs 5th generation warfare (2)
3 MIN READ
0
NGS vs 5th generation warfare (1)
3 MIN READ
0
Not necessary. Not safe. Not effective (10)
2 MIN READ
0
Not necessary. Not safe. Not effective (9)
3 MIN READ
0
Not necessary. Not safe. Not effective (8)
< 1 MIN READ
0
Not necessary. Not safe. Not effective (7)
2 MIN READ
0
Not necessary. Not safe. Not effective (4)
2 MIN READ
0
Not necessary. Not safe. Not effective (2)
2 MIN READ
0
Not necessary. Not safe. Not effective (1)
3 MIN READ
0
miRNA-mediated predictions (10)
3 MIN READ
0
miRNA-mediated predictions (9)
2 MIN READ
0
miRNA-mediated predictions (8)
2 MIN READ
0
miRNA-mediated preditions (7)
3 MIN READ
0
miRNA-mediated predictions (6)
2 MIN READ
0
miRNA-mediated predictions (5)
3 MIN READ
0
miRNA-mediated predictions (4)
2 MIN READ
0
miRNA-mediated predictions (3)
< 1 MIN READ
0
miRNA-mediated predictions (2)
2 MIN READ
0
miRNA-mediated preditions (1)
< 1 MIN READ
0
Minimal level of conflict (10)
5 MIN READ
0
Minimal level of conflict (9)
2 MIN READ
0
Minimal level of conflict (8)
4 MIN READ
0
Minimal level of conflict (7)
2 MIN READ
0
Minimal level of conflict (6)
2 MIN READ
0
Minimal level of conflict (5)
2 MIN READ
0
Minimal level of conflict (4)
2 MIN READ
0
Minimal level of conflict (3)
3 MIN READ
0
Minimal level of conflict (2)
2 MIN READ
0
Minimal level of conflict (1)
4 MIN READ
0
Biorealism (10)
< 1 MIN READ
0
Biorealism (9)
< 1 MIN READ
0
Biorealism (8)
2 MIN READ
0
Biorealism (7)
< 1 MIN READ
0
Biorealism (6)
2 MIN READ
0
Biorealism (5)
4 MIN READ
0
Biorealism (4)
2 MIN READ
0
Biorealism (2)
3 MIN READ
0
Biorealism (1)
2 MIN READ
0
Total recall 6000 years (10)
2 MIN READ
0
Total recall 6000 years (9)
2 MIN READ
0
Total recall 6000 years (8)
2 MIN READ
0
Total recall 6000 years (7)
2 MIN READ
0
Total recall 6000 years (6)
2 MIN READ
0
Total recall 6000 years (5)
2 MIN READ
0
Total recall 6000 years (4)
3 MIN READ
0
Total recall 6000 years (3)
2 MIN READ
0
Total recall 6000 years (2)
< 1 MIN READ
0
Total recall 6000 years (1)
3 MIN READ
0
Censorship of perception (10)
< 1 MIN READ
0
Censorship of perception (9)
< 1 MIN READ
0
Censorship of perception (8)
2 MIN READ
0
Censorship of perception (7)
2 MIN READ
0
Censorship of perception (6)
2 MIN READ
0
Censorship of perception (5)
3 MIN READ
0
Censorship of perception (4)
< 1 MIN READ
0
Censorship of perception (3)
3 MIN READ
0
Censorship of perception (2)
3 MIN READ
0
Censorship of perception (1)
2 MIN READ
0
Biochemical is geopolitical (10)
2 MIN READ
0
Biogeochemical is geopolitical (9)
2 MIN READ
0
Biogeochemical is geopolitical (8)
2 MIN READ
0
Biogeochemical is geopolitical (7)
2 MIN READ
0
Biogeochemical is geopolitical (6)
2 MIN READ
0
Biogeochemical is geopolitical (4)
3 MIN READ
0
Biogeochemical is geopolitical (3)
2 MIN READ
0
Biogeochemical is geopolitical (2)
3 MIN READ
0
Biogeochemical is geopolitical (1)
2 MIN READ
0
Flipping off the flipons (10)
4 MIN READ
0
Flipping off the flipons (9)
< 1 MIN READ
0
Flipping off the flipons (8)
< 1 MIN READ
0
Flipping off the flipons (7)
< 1 MIN READ
0
Flipping off the flipons (6)
2 MIN READ
0
Flipping off the flipons (5)
2 MIN READ
0
Flipping off the flipons (4)
4 MIN READ
0
Flipping off the flipons (3)
3 MIN READ
0
Flipping off the flipons (2)
< 1 MIN READ
0
Patenting the sun (10)
2 MIN READ
0
Patenting the sun (9)
< 1 MIN READ
0
Patenting the sun (8)
7 MIN READ
0
Patenting the sun (7)
3 MIN READ
0
Patenting the sun (6)
4 MIN READ
0
Patenting the sun (5)
3 MIN READ
0
Patenting the sun (4)
2 MIN READ
0
Patenting the sun (3)
2 MIN READ
0
Patenting the sun (2)
3 MIN READ
0
Patenting the sun (1)
< 1 MIN READ
0
Positive as a proton (10)
2 MIN READ
0
Positive as a proton (9)
2 MIN READ
0
Positive as a proton (8)
2 MIN READ
0
Positive as a proton (7)
2 MIN READ
0
Positive as a proton (6)
< 1 MIN READ
0
Positive as a proton (4)
3 MIN READ
0
Positive as a proton (3)
3 MIN READ
0
Positive as a proton (2)
< 1 MIN READ
0
Positive as a proton (1)
2 MIN READ
0
WHO broke the breakthrough (10)
3 MIN READ
0
WHO broke the breakthough (9)
3 MIN READ
0
WHO broke the breakthrough (8)
< 1 MIN READ
0
WHO broke the breakthrough (7)
< 1 MIN READ
0
WHO broke the breakthrough (6)
2 MIN READ
0
WHO broke the breakthrough (3)
6 MIN READ
0
WHO broke the breakthrough (2)
2 MIN READ
0
WHO broke the breakthrough (1)
3 MIN READ
0
miRNA-mediated epigenetic effects (10)
3 MIN READ
0
miRNA-mediated epigenetic effects (9)
2 MIN READ
0
miRNA-mediated epigenetic effects (8)
< 1 MIN READ
0
miRNA-mediated epigenetic effects (7)
< 1 MIN READ
0
miRNA-mediated epigenetic effects (6)
2 MIN READ
0
miRNA-mediated epigenetic effects (5)
6 MIN READ
0
miRNA-mediated epigenetic states (3)
2 MIN READ
0
miRNA-mediated epigenetic states (2)
2 MIN READ
0
The devil is in the dirt (10)
4 MIN READ
0
The devil is in the dirt (9)
< 1 MIN READ
0
The devil is in the dirt (8)
2 MIN READ
0
The devil is in the dirt (7)
< 1 MIN READ
0
The devil is in the dirt (6)
2 MIN READ
0
The devil is in the dirt (5)
2 MIN READ
0
The devil is in the dirt (4)
2 MIN READ
0
The devil is in the dirt (3)
5 MIN READ
0
The devil is in the dirt (2)
2 MIN READ
0
The devil is in the dirt (1)
2 MIN READ
0
Scientism, atheism and theology (10)
2 MIN READ
0
Scientism, Atheism and Theology (9)
2 MIN READ
0
The tipping point (revisited): 151K
2 MIN READ
0
Scientism, Atheism and Theology (7)
< 1 MIN READ
0
Scientism, Atheism & Theology (6)
2 MIN READ
0
Scientism, Atheism & Theology (4)
2 MIN READ
0
Scientism, Atheism & Theology (3)
< 1 MIN READ
0
Scientism, Atheism & Theology (2)
2 MIN READ
0
Pheromone regulated genetic processes (9)
2 MIN READ
0
Pheromone-regulated genetic processes (8)
2 MIN READ
0
Pheromone-regulated genetic processes (7)
< 1 MIN READ
0
Pheromone-regulated genetic processes (4)
2 MIN READ
0
Pheromone-regulated genetic processes (1)
2 MIN READ
0
The physics of existence (9)
3 MIN READ
0
The physics of existence (6)
2 MIN READ
0
The physics of existence (5)
2 MIN READ
0
The physics of existence (4)
3 MIN READ
0
Separated we survive (1)
3 MIN READ
0
Biophotonically charged life (5)
2 MIN READ
0
Biophotonically charged life (4)
2 MIN READ
0
Biophotonically charged life (3)
< 1 MIN READ
0
Codon optimality vs systemic fraud (10)
3 MIN READ
0
Codon optimality vs systemic fraud (9)
5 MIN READ
0
Codon optimality vs systemic fraud (7)
3 MIN READ
0
Codon optimality vs systemic fraud (3)
2 MIN READ
0
Codon optimality vs systemic fraud (1)
6 MIN READ
0
Epistemic corruption (8)
2 MIN READ
0
Epistemic corruption (3)
2 MIN READ
0
Epistemic corruption (1)
3 MIN READ
0
MicroRNA-mediated existential threats (8)
4 MIN READ
0
MicroRNA-mediated existential threats (3)
4 MIN READ
0
MicroRNA-mediated existential threats (1)
4 MIN READ
0
RNA-mediated silencing (9)
4 MIN READ
0
RNA-mediated silencing (8)
2 MIN READ
0
RNA-mediated silencing (7)
3 MIN READ
0
RNA-mediated silencing (3)
3 MIN READ
0
Photonics in Forensics (10)
3 MIN READ
0
Photonics in Forensics (9)
6 MIN READ
0
Photonics in Forensics (8)
4 MIN READ
0
Photonics in Forensics (7)
3 MIN READ
0
Photonics in Forensics (6)
2 MIN READ
0
Photonics in Forensics (1)
2 MIN READ
0
microRNA-mediated biodiversity (10)
3 MIN READ
0
microRNA-mediated biodiversity (9)
< 1 MIN READ
0
microRNA-mediated biodiversity (6)
3 MIN READ
0
microRNA-mediated biodiversity (5)
2 MIN READ
0
microRNA-mediated biodiversity (4)
2 MIN READ
0
microRNA-mediated biodiversity (2)
2 MIN READ
0
microRNA-mediated biodiversity (1)
2 MIN READ
0
“The Darwin Code: Intelligent Design without God” (4)
3 MIN READ
0
“The Darwin Code: Intelligent Design without God” (3)
< 1 MIN READ
0
microRNA-mediated cures (8)
4 MIN READ
0
Naturally attenuated viral endemicity (8)
3 MIN READ
0
Naturally attenuated viral endemicity (4)
2 MIN READ
0
Naturally attenuated viral endemicity (1)
4 MIN READ
0
Orchestrated death: Francis S. Collins (2)
4 MIN READ
0
Orchestrated death: Sir Paul Nurse
2 MIN READ
0
Natural selection for adaptation (2)
2 MIN READ
0
Wars caused by metaphors: 1964
3 MIN READ
0
MicroRNA-mediated population control (3)
3 MIN READ
0
MicroRNA-mediated population control (2)
3 MIN READ
0
microRNA-mediated election cycles (6)
3 MIN READ
0
microRNA-mediated replication cycles (6)
2 MIN READ
0
microRNA-mediated replication timing (5)
3 MIN READ
0
The tipping point (revisited): 120K
2 MIN READ
0
Quantum Darwinism (6)
2 MIN READ
0
Pheromones protect us from viruses (3)
3 MIN READ
0
Defeating disease and Communism (4)
2 MIN READ
0
Pheromones protect us from viruses (2)
1 MIN READ
0
Impeaching the God of Abraham (9)
2 MIN READ
0
Impeaching the God of Abraham (8)
4 MIN READ
0
Impeaching the God of Abraham (10)
< 1 MIN READ
0
Life or death facts about theories (4)
3 MIN READ
0
Life or death facts about theories (3)
2 MIN READ
0
Creationism, Communism and Corruption (1)
2 MIN READ
0
God’s protection from SARS COV-2 and other viruses (3)
2 MIN READ
0
Racist fascism vs individual liberty (1)
3 MIN READ
0
Protonated RNA interference vs stupid theories (4)
< 1 MIN READ
0
Autophagy-related microRNA-mediated disease treatment (1)
4 MIN READ
0
pH-dependent viral latency (5)
4 MIN READ
0
pH-dependent viral latency (4)
3 MIN READ
0
pH-dependent viral latency (3)
4 MIN READ
0
pH-dependent viral latency (2)
3 MIN READ
0
The tipping point (revisited): 102K (1)
2 MIN READ
0
The microRNA-mediated future of humanity (3)
3 MIN READ
0
FDA’s ignorance: From roots to shoots (2)
3 MIN READ
0
CDC’s ignorance: from roots to shoots (3)
4 MIN READ
0
The tipping point (revisited): 100K (2)
5 MIN READ
0
The tipping point (revisited): 100K (1)
4 MIN READ
0
NIH’s ignorance: From roots to shoots (1)
4 MIN READ
0
FDA’s ignorance: From roots to shoots (1)
6 MIN READ
0
From Wuhan to you (4)
2 MIN READ
0
Richard Axel refutes theistic evolution
2 MIN READ
0
Viral microRNA-mediated pathology (2)
3 MIN READ
0
MicroRNA-mediated healthy longevity (3)
3 MIN READ
0
Bruce McEwen’s legacy: sympatric speciation (5)
4 MIN READ
0
The tipping point (revisited): 96K (1)
2 MIN READ
0
A microRNA-mediated healthy New Year! Thank God (1)
4 MIN READ
0
The end of theistic evolution (1)
2 MIN READ
0
microRNA-mediated prevention of diseases (1)
2 MIN READ
0
microRNA-mediated sex differences (1)
2 MIN READ
0
The tipping point (revisited): 95K (5)
< 1 MIN READ
0
The tipping point (revisited): 95K (2)
2 MIN READ
0
The tipping point (revisited): 95K (1)
< 1 MIN READ
0
Biological Function of Autophagy (2)
7 MIN READ
0
Biological Function of Autophagy (1)
2 MIN READ
0
MicroRNAs biophysically constrain Virus-driven pathology (6)
3 MIN READ
0
MicroRNAs biophysically constrain Virus-driven pathology (5)
4 MIN READ
0
Virus-driven sympatric speciation (1)
2 MIN READ
0
How to Profit from Suffering and Death (1)
2 MIN READ
0
MicroRNAs on Veteran’s Day 2019
< 1 MIN READ
0
MicroRNA-mediated hypoxia vs cancer (1)
5 MIN READ
0
The tipping point (revisited): 93K (1)
4 MIN READ
0
2020 National Vaccine Plan (1)
2 MIN READ
0
#SFN2019 to 2012: Hiding the facts (7)
2 MIN READ
0
#SFN2019 to 2012: Hiding the facts (3)
3 MIN READ
0
#SFN2019 to 2012: Hiding the facts (2)
6 MIN READ
0
#SFN2019 to 2012: Hiding the facts (1)
3 MIN READ
0
Multigenerational epigenetic inheritance (3)
3 MIN READ
0
Patented Creation vs Evolution of Disease (9)
4 MIN READ
0
Patented Creation vs Evolution of Disease (8)
3 MIN READ
0
Patented Creation vs Evolution of Disease (6)
4 MIN READ
0
Patented Creation vs Evolution of Disease (2)
3 MIN READ
0
Patented Creation vs Evolution of Disease (1)
9 MIN READ
0
Chemogenetic kinetics (7): DHA vs consensus (4)
3 MIN READ
0
Chemogenetic kinetics (7): DHA vs consensus (3)
3 MIN READ
0
Light-activated chemogenetic kinetics (2)
4 MIN READ
0
MicroRNA-mediated, since 1964 (1)
2 MIN READ
0
Chemogenetic kinetics (2): Extinction
6 MIN READ
0
Chemogenetic kinetics (1): Creation
3 MIN READ
0
Nutrient-dependent Pheromone-controlled cures (3)
4 MIN READ
0
Lewis Thomas (revisited): 90K (7)
3 MIN READ
0
Brokered regulatory leverage: Breaking Bad (1)
5 MIN READ
0
Sunlight, hydrophobicity and biodiversity (3)
5 MIN READ
0
The tipping point (revisited): 90K (1)
4 MIN READ
0
Bastardized health care (4)
4 MIN READ
0
Weaponized health information (6)
6 MIN READ
0
Weaponized health information (8)
6 MIN READ
0
Weaponized health communication (5)
2 MIN READ
0
Weaponized health communication (3)
4 MIN READ
0
Genetic endemism: apologetics vs epigenetics (1)
2 MIN READ
0
Codon-dependent mRNA stability (2)
2 MIN READ
0
Relatable not debatable (1)
4 MIN READ
0
Food energy-as-information, and consciousness
4 MIN READ
0
pH, amino acids, and health: Effects of sunlight and well wishes
2 MIN READ
0
The tipping point (revisited): 88K (3)
2 MIN READ
0
Abiogenesis vs microRNA biogenesis (3)
3 MIN READ
0
The tipping point (revisited): 87K (3)
4 MIN READ
0
The eternal significance of microRNA biogenesis (3)
3 MIN READ
0
The tipping point (revisited): Big Data (2)
2 MIN READ
0
The tipping point (revisited): HYL1
3 MIN READ
0
Tasting light links energy from creation to adaptation (3)
2 MIN READ
0
Ecological Adaptations: From Angstroms to Ecosystems (2)
3 MIN READ
0
Ecological Adaptations: From Angstroms to Ecosystems (1)
4 MIN READ
0
Tasting light links energy from creation to adaptation (2)
3 MIN READ
0
Kohl and Francoeur at 25 (2)
2 MIN READ
0
Gamification of creationism (1)
4 MIN READ
0
Biophysical constraints on RNA synthesis (1)
9 MIN READ
0
Virus-driven downsizing of the human brain (6)
6 MIN READ
0
DS Wilson’s view of biophysically constrained life (3)
3 MIN READ
0
DS Wilson’s view of biophysically constrained life (2)
2 MIN READ
0
RNA processing and disease (3)
3 MIN READ
0
Ecological adaptation vs neo-Darwinian nonsense (1)
2 MIN READ
0
Non-random vs random selection for receptor clonotypes
3 MIN READ
0
Blood music orchestrates human life (3)
3 MIN READ
0
Blood music orchestrates human life (1)
5 MIN READ
0
Do not call me Ishmael (2)
2 MIN READ
0
Biophysically constrained fast adaptation (3)
5 MIN READ
0
10,000 reasons to believe in biophysical constraints (revisited)
< 1 MIN READ
0
Phytoremediation, Microbiome and CRISPR (3)
5 MIN READ
0
Autophagy automagically prevents cancer (2)
2 MIN READ
0
Phytoremediation, Microbiome and CRISPR (1)
3 MIN READ
0
Light-activated continuous environmental tracking (2)
3 MIN READ
0
Light-activated continuous environmental tracking (1)
5 MIN READ
0
Light and life at base pair resolution (10)
4 MIN READ
0
Dating rocks or mating for biodiversity?
6 MIN READ
0
Life and death at base pair resolution (1)
< 1 MIN READ
0
Light and life at base pair resolution (9)
4 MIN READ
0
Code Biology vs Predatory Publishing (2)
2 MIN READ
0
Two photon-linked biodiversity (3)
2 MIN READ
0
The tipping point (revisited): 81,000 publications (2)
2 MIN READ
0
The tipping point (revisited): 81,000 publications (1)
5 MIN READ
0
From microRNA.pro to quantumsouls.pro (2)
3 MIN READ
0
Light and life at base pair resolution (1)
2 MIN READ
0
Hide and seek with science facts (7)
3 MIN READ
0
One mutation vs two amino acid substitutions
3 MIN READ
0
Bottom-up biology
3 MIN READ
0
Electrons build fractal shapes and prevent lung cancer
2 MIN READ
0
SfN’s 2018 trap set for Democrats
2 MIN READ
0
Evolved suffering and death caused by statistical significance
< 1 MIN READ
0
War Games: False Flag Terrorism (4)
3 MIN READ
0
Creating an enzyme that kills theories (2)
6 MIN READ
0
Creating an enzyme that kills theories (1)
< 1 MIN READ
0
Emergence of a coherent model
< 1 MIN READ
0
The tipping point (revisited): 78,000 publications (3)
< 1 MIN READ
0
Theorists: The biggest threat to microRNA-mediated security
5 MIN READ
0
Epigenetic effects on Eastern and Western societies
2 MIN READ
0
Michael Bloomberg for President (NOT) in 2020 (3)
< 1 MIN READ
0
Michael Bloomberg for President (NOT) in 2020
2 MIN READ
0
Energy-dependent ‘futile cycles’ of autophagy
2 MIN READ
0
Quantized energy-dependent microRNA-mediated autophagy (2)
< 1 MIN READ
0
Natural models inspire future therapeutics (2)
2 MIN READ
0
Quantum initiation of cold chemistry vs Hypeology (3)
3 MIN READ
0
Quantum initiation of cold chemistry vs Hypeology (2)
3 MIN READ
0
Estranged ‘White Coat Notes’ blogger exposes human idiocy (2)
3 MIN READ
0
Estranged ‘White Coat Notes’ blogger exposes human idiocy (1)
5 MIN READ
0
Quantum initiation of cold chemistry vs Hypeology (1)
4 MIN READ
0
Virus-driven genome engineering causes cancer?
2 MIN READ
0
The concept of a species (4)
2 MIN READ
0
The concept of a species (3)
4 MIN READ
0
The concept of a species (1)
2 MIN READ
0
The tipping point (revisited): 74,000 publications
4 MIN READ
0
Laws of Biology vs no Laws of Physics (2)
3 MIN READ
0
MicroRNA-mediated alternative splicing (revisited)
4 MIN READ
0
Corporate owned microRNA-mediated biodiversity
< 1 MIN READ
0
Animal welfare vs human ethology
4 MIN READ
0
Biologically uninformed science idiot: Self-defense (4)
4 MIN READ
0
Biologically uninformed science idiot: Self-defense (3)
5 MIN READ
0
Biologically uninformed science idiot: Self-defense (2)
3 MIN READ
0
The eternal significance of microRNAs and the Vietnam Memorial (1)
3 MIN READ
0
Complexity: Routes and Patterns (5)
5 MIN READ
0
From quantum physics to quantum souls (2)
2 MIN READ
0
Epigenetic inheritance of spatiotemporal regulation (3)
5 MIN READ
0
Epigenetic inheritance of spatiotemporal regulation
4 MIN READ
0
The origin of information (5,4,3,2,1)
3 MIN READ
0
The tipping point (revisited): 73,000 publications
5 MIN READ
0
The eternal significance of microRNAs (8)
2 MIN READ
0
The eternal significance of microRNAs (7)
3 MIN READ
0
The eternal significance of microRNAs (5)
2 MIN READ
0
ATP and RNA-mediated chromosomal stability
3 MIN READ
0
A single base change refutes theistic evolution (2)
2 MIN READ
0
Ecological adaptations vs the randomness of evolution (3)
4 MIN READ
0
EDAR V370A and sympatric speciation
2 MIN READ
0
Ecological adaptations vs the randomness of evolution (4)
< 1 MIN READ
0
Environmental selection is natural selection (5)
5 MIN READ
0
Environmental selection is natural selection (4)
2 MIN READ
0
Environmental selection is natural selection (2)
2 MIN READ
0
Environmental selection is natural selection
3 MIN READ
0
MicroRNA-mediated denuclearization (6)
2 MIN READ
0
Abiogenesis vs microRNA biogenesis (2)
7 MIN READ
0
Abiogenesis vs microRNA biogenesis
4 MIN READ
0
2018 March for Science vs microRNAs
7 MIN READ
0
The eternal significance of microRNAs (3)
3 MIN READ
0
The eternal significance of microRNAs (1)
< 1 MIN READ
0
Sympatric Speciation vs pseudosceintific nonsense (4)
2 MIN READ
0
Sympatric Speciation vs Pseudoscientific Nonsense (1)
4 MIN READ
0
Complexity: Routes and Patterns (4)
< 1 MIN READ
0
Laws of Biology / Rules of Life
3 MIN READ
0
Less evolvable RNA-mediated ecological adaptations
3 MIN READ
0
Psychophysical Laws of Biology: RNA-mediated nutritional psychiatry (3)
4 MIN READ
0
Odor activation of ATP (2)
2 MIN READ
0
Part 3: Light-controlled cell biology (revisited)
3 MIN READ
0
Light-controlled cell biology (revisited)
6 MIN READ
0
Death cures theorist’s Amyotrophic Lateral Sclerosis (ALS).
< 1 MIN READ
0
Polymaths and paradigm shifts: From Asimov to Bear (5)
5 MIN READ
0
Polymaths and paradigm shifts: from Asimov to Bear (4)
6 MIN READ
0
Polymaths and paradigm shifts: from Asimov to Bear (3)
3 MIN READ
0
Polymaths and paradigm shifts: from Asimov to Bear (2)
4 MIN READ
0
Polymaths and paradigm shifts: from Asimov to Bear (1)
5 MIN READ
0
How to create biologically uninformed theorists
7 MIN READ
0
Ecological adaptation: a new definition of heredity (4)
4 MIN READ
0
Ecological adaptation: A new definition of heredity (3)
4 MIN READ
0
MicroRNAs biophysically constrain behavior (2)
3 MIN READ
0
Conceptual critique: Innateness vs the death gene (1)
5 MIN READ
0
How to biophysically constrain the flu virus (1)
4 MIN READ
0
Diet-driven RNA interference and mental health (4)
8 MIN READ
0
Subatomic: From thermophiles to humans (3)
3 MIN READ
0
Cryo-EM: Linking spatial and conformational constraints
9 MIN READ
0
Subatomic: From thermophiles to humans (2)
6 MIN READ
0
Subatomic: From thermophiles to humans
5 MIN READ
0
A reversible TCA cycle in a thermophile (3)
4 MIN READ
0
A reversible TCA cycle in a thermophile (2)
3 MIN READ
0
A reversible TCA cycle in a thermophile
5 MIN READ
0
A mental health problem at the highest level (3)
7 MIN READ
0
Diet-driven RNA interference and mental health (3)
4 MIN READ
0
Anti-entropic sunlight: Schrödinger’s Creationist Secret? (3)
6 MIN READ
1
The MicroRNAome Strikes Back: A Sokalian hoax (10)
3 MIN READ
0
BiondVax Universal Flu Vaccine Patent
4 MIN READ
0
The MicroRNAome Strikes Back: A Sokalian hoax (8)
4 MIN READ
0
The MicroRNAome Strikes Back: A Sokalian hoax (4)
5 MIN READ
0
The MicroRNAome Strikes Back: A Sokalian hoax (3)
4 MIN READ
0
The MicroRNAome Strikes Back: A Sokalian hoax (2)
6 MIN READ
0
The tipping point (revisted): 69,000 publications
4 MIN READ
0
A mental health problem at the highest level (2)
3 MIN READ
0
Diet-driven RNA interference and cancer prevention (3)
6 MIN READ
0
Autophagy is the antiphage defense strategy (2)
4 MIN READ
0
Enzyme-constrained interethnic diversity (8)
< 1 MIN READ
0
Enzyme-constrained interethnic diversity (7)
6 MIN READ
0
Diet-driven RNA interference and cancer prevention
5 MIN READ
0
Enzyme-constrained interethnic biodiversity (5)
4 MIN READ
0
Enzyme-constrained interethnic biodiversity (3)
3 MIN READ
0
Enzyme-constrained interethnic biodiversity (1)
5 MIN READ
0
Quantized energy-dependent viral trophism (2)
2 MIN READ
0
A Mathematical Model Links Quantum Physics to Quantum Souls (3)
5 MIN READ
0
A Mathematical Model Links Quantum Physics to Quantum Souls (2)
4 MIN READ
0
The “walking fish” walks straight from quantum physics to quantum souls (4)
11 MIN READ
0
The “walking fish” walks straight from quantum physics to quantum souls (2)
3 MIN READ
0
A Mathematical Model Links Quantum Physics to Quantum Souls (1)
6 MIN READ
0
From base editing to RNA editing (8)
4 MIN READ
0
From base editing to RNA editing (6)
8 MIN READ
0
Who created your virus-driven death gene? (3)
3 MIN READ
0
Kinetically Stable Thermodynamically Activated Cell Metabolism (7)
4 MIN READ
0
Kinetically Stable Thermodynamically Activated Cell Metabolism (5)
4 MIN READ
0
Kinetically Stable Thermodynamically Activated Cell Metabolism (4)
3 MIN READ
0
Kinetically Stable Thermodynamically Activated Cell Metabolism (3)
3 MIN READ
0
Kinetically Stable Thermodynamically Activated Cell Metabolism (2)
4 MIN READ
0
Kinetically Stable Thermodynamically Activated Cell Metabolism (1)
3 MIN READ
0
The tipping point (revisited): 68,000 publications
5 MIN READ
0
Agilent technology and energy-dependent autophagy
8 MIN READ
0
Tai Chi vs PTSD and cancer
< 1 MIN READ
0
Trump challenges the CDC to remember 9/11
14 MIN READ
0
Narcissistic egomaniacal and deadly denial of autophagy
6 MIN READ
0
Elsevier fails to support the concept of autophagy
4 MIN READ
0
From Autophagy.pro (transitions)
4 MIN READ
0
Nature vs Science and Autophagy.pro
7 MIN READ
0
The overwhelming ignorance of sex researchers (2)
4 MIN READ
0
Who created your virus-driven death gene? (2)
10 MIN READ
0
Energy-dependent base editing and correction of mutations (2)
9 MIN READ
0
Trashing the 2nd Law
7 MIN READ
0
The overwhelming ignorance of sex researchers
5 MIN READ
0
Mouse morphs and primate diversity in 50 years
7 MIN READ
0
Two retractions of human idiocy
2 MIN READ
0
Exposing the 2nd Law to more ridicule (1)
6 MIN READ
0
Vulgar Antagonists vs Quantum Effects
3 MIN READ
0
Reporting new scientific truths is not allowed in the USA
3 MIN READ
0
Who created your virus-driven death gene? (1)
5 MIN READ
0
Denying Creation via RNA-directed DNA methylation
4 MIN READ
0
Light-activated error free DNA repair (2)
8 MIN READ
0
Schrodinger’s answer to Schrodinger’s question
3 MIN READ
0
Light-activated error-free DNA repair (4)
3 MIN READ
0
MicroRNAs and the Cassandra syndrome (revisited)
4 MIN READ
0
A null mutation and healthy longevity?
4 MIN READ
0
Light-activated error free DNA repair
5 MIN READ
0
Achromobacter and killer theories
2 MIN READ
0
Eutrophication and phosphorylated biodiversity
6 MIN READ
0
Alternative splicing: a new therapy?
3 MIN READ
0
Epigenetic facts vs variable recombination theories
4 MIN READ
0
Selfish X-linked alleles and X chromosome inactivation
3 MIN READ
0
Pheromones biophysically constrain RNA-mediated biodiversity (1)
5 MIN READ
0
Pheromones biophysically constrain base editing and RNA editing
3 MIN READ
0
From base editing to RNA editing (5)
< 1 MIN READ
0
From base editing to RNA editing (4)
5 MIN READ
0
The next reformation: 500 years too late? (2)
2 MIN READ
0
The next reformation: 500 years too late? (3)
5 MIN READ
0
From base editing to RNA editing (2)
7 MIN READ
0
From base editing to RNA editing
2 MIN READ
0
Loss of energy as information (1)
3 MIN READ
0
Cryo-EM: More than a suggestion
< 1 MIN READ
0
Long-term adaptation replaces evolution (3)
6 MIN READ
0
Long-term adaptation replaces evolution (2)
2 MIN READ
0
Exome Sequencing Impact in Routine Care
4 MIN READ
0
Losers: rediscovering an epigenetic key
2 MIN READ
0
Virulence: Out of stock
2 MIN READ
0
Faith in evolution: Paleontology as a molecular science
5 MIN READ
0
Energy-dependent structure and function: Until death (5)
4 MIN READ
0
Energy-dependent structure and function: Until death (3)
3 MIN READ
0
Energy-dependent structure and function: Until death (2)
5 MIN READ
0
Energy-dependent structure and function: Until death (1)
4 MIN READ
0
Biophysically constrained intelligence vs human idiocy
4 MIN READ
0
Cryo-EM from atoms to ecosystems
6 MIN READ
0
Predicting who wins the 2017 Nobel Prizes (7)
4 MIN READ
0
Until death: Virus-driven failure of multisensory integration (1)
4 MIN READ
0
Entertaining yourself to death
6 MIN READ
0
Criticisms of a validated model
9 MIN READ
0
Predicting who wins the 2017 Nobel Prizes (5)
2 MIN READ
0
Predicting who wins the 2017 Nobel Prizes (4)
4 MIN READ
0
Predicting who wins the 2017 Nobel Prizes (2)
4 MIN READ
0
Predicting who wins the 2017 Nobel Prizes
4 MIN READ
0
Life and death via chemical effects on photosynthesis
8 MIN READ
0
Dispelling the ignorance of theorists
4 MIN READ
0
Sci-Hub vs limits on the dissemination of information
6 MIN READ
0
Evolutionary theories of epigenetic drift (2)
5 MIN READ
0
Evolutionary theories of epigenetic drift
2 MIN READ
0
Evolution outside the context of “the light of evolution” (2)
3 MIN READ
0
Evolution outside the context of “the light of evolution”
6 MIN READ
0
Inventing “Transcriptome Trajectory Turning Points”
2 MIN READ
0
Optogenetics replaces fMRI but not pseudoscientific nonsense
2 MIN READ
0
Science vs Secularism: Molecular Mechanisms or Math?
6 MIN READ
0
Methylation and the Innate Immune System
< 1 MIN READ
0
MicroRNAs: Nature’s secret ingredient
2 MIN READ
0
Sexual communication signals: New Insights!
10 MIN READ
0
Host-derived creation of all pathology (2 of 2)
9 MIN READ
0
Host-derived creation of all pathology (1 of 2)
5 MIN READ
0
An alternative mode of bacterial quorum sensing?
3 MIN READ
0
Human Idiocy: The failure to link energy from electrons to ecosystems
19 MIN READ
0
Stem cells do not create themselves
3 MIN READ
0
Pseudoscientists hate what science explains! (3)
4 MIN READ
0
Pseudoscientists hate what science explains!
3 MIN READ
0
Your indifference is killing you and others
5 MIN READ
0
Your indifference is killing you and others (4)
4 MIN READ
0
Robert Sapolsky’s legacy of atheistic pseudoscientific nonsense
4 MIN READ
0
Automagical protein creation sans energy
9 MIN READ
0
Sunlight, phytochemicals, microRNAs and cancer
5 MIN READ
0
Solar analemma with a total solar eclipse
2 MIN READ
0
The Origin of Information (4)
4 MIN READ
0
Can protein folding chemistry be understood by theorists?
5 MIN READ
0
Evolutionary congruence (losers) vs ecological adaptation (winners)
3 MIN READ
0
The Origin of Information (3)
6 MIN READ
0
Irreconcilable differences: food energy vs de novo assembly
2 MIN READ
0
Pattern recognition vs evolutionary processes (revisited)
7 MIN READ
0
New Testament Book Cover Refutes Theistic Evolution
7 MIN READ
0
Measuring energy vs human idiocy (social science)
2 MIN READ
0
Energy-dependent physical and biophysical constraints (10)
8 MIN READ
0
Energy-dependent physical and biophysical constraints (9)
3 MIN READ
0
Energy-dependent physical and biophysical constraints (8)
5 MIN READ
0
Energy-dependent physical and biophysical constraints (6)
8 MIN READ
0
Energy-dependent physical and biophysical constraints (5)
7 MIN READ
0
Energy-dependent physical and biophysical constraints (3)
2 MIN READ
0
Energy-dependent physical and biophysical constraints (2)
4 MIN READ
0
Energy-dependent physical and biophysical constraints (1)
5 MIN READ
0
Cell type assembly and the space-time continuum
2 MIN READ
0
Richard P. Feynman refuted theistic evolution
5 MIN READ
0
Epigenetic effects of stress by Bruce McEwen (2)
3 MIN READ
0
The emergence of light as energy from a life-giving star
11 MIN READ
2
God’s shrinking role in salvation (3)
9 MIN READ
0
God's shrinking role in salvation (3)
9 MIN READ
0
MicroRNAs GnRH and the failure of sex research
7 MIN READ
0
God’s shrinking role in salvation (2)
5 MIN READ
0
God’s shrinking role in salvation
8 MIN READ
0
Food energy-dependent cell type differentiation (2)
10 MIN READ
0
Hard Science vs the neo-Darwinian “magic box”
9 MIN READ
0
MicroRNAs light up life
7 MIN READ
0
Infections and age change the odor of wine and people
4 MIN READ
0
From E. coli to monkeys and mankind: Theories vs models (2)
6 MIN READ
0
From E. coli to monkeys and mankind: Theories vs models
7 MIN READ
0
Food energy-dependent epigenetic adaptation (3)
3 MIN READ
0
Food energy-dependent epigenetic adaptation (2)
< 1 MIN READ
0
Food energy-dependent epigenetic adaptation
11 MIN READ
0
Energy-dependent epigenetic translation to mRNA stability (6)
8 MIN READ
0
Energy-dependent epigenetic translation to mRNA stability (5)
4 MIN READ
0
Energy-dependent epigenetic translation to mRNA stability (3)
5 MIN READ
0
Energy-dependent epigenetic translation to mRNA stability
8 MIN READ
0
UV light and non-coding RNA
2 MIN READ
0
Energy as information and constrained endogenous RNA interference (4)
7 MIN READ
0
Energy-dependent microRNA biogenesis
4 MIN READ
0
Draining the academic swamp of parasites
3 MIN READ
0
The death of human ethology via ecology
7 MIN READ
0
Dispensing with all pseudoscientific nonsense about evolution (2)
< 1 MIN READ
0
Dispensing with all pseudoscientific nonsense about evolution (1)
5 MIN READ
0
Virus-driven downsizing of the human brain (4)
2 MIN READ
0
Feng Zhang refutes theistic evolution
4 MIN READ
0
Open Science: Closed to facts about microRNAs
6 MIN READ
0
Viruses in pathogenic variants disrupt alternative splicings (2)
3 MIN READ
0
Viruses in pathogenic variants disrupt alternative splicings
2 MIN READ
0
Respiration-dependent endogenous RNA interference
4 MIN READ
0
Epigenetically-effected nucleosome repositioning sheds Dobzhansky’s light on evolution
3 MIN READ
0
Cytosis: Gameplay Runthrough
3 MIN READ
0
Synthetic RNA-based switches
2 MIN READ
0
Sunlight bursts the social bubble of physics and math
3 MIN READ
0
Functionally interdependent editing and methylation
4 MIN READ
0
Cytosis: Biology Content
7 MIN READ
0
Light-activated feedback loops vs self-organization of ecosystems
7 MIN READ
0
Harvard researchers support young earth creationism
< 1 MIN READ
0
Cytosis: A Cell Biology Board Game
< 1 MIN READ
0
Sal Giardina: apologetics revisited
8 MIN READ
0
Life in your UV light-constrained galaxy
5 MIN READ
0
Food for thought: Penrose v Ellis (3)
4 MIN READ
0
Thinking about energy is not radical re-thinking
6 MIN READ
0
Magnussen’s “Faith fools” keep the faith
3 MIN READ
0
Energy-dependent pheromone-controlled entropy (3)
2 MIN READ
0
Energy-dependent pheromone-controlled entropy (4)
3 MIN READ
0
Proof: Sunlight is energy as information
< 1 MIN READ
0
Energy-dependent pheromone-controlled entropy (2)
6 MIN READ
0
Sunlight: the essence of precision medicine and world peace
6 MIN READ
0
The essence of precision medicine: drug targets or healthy longevity?
4 MIN READ
0
The essence of precision medicine
4 MIN READ
0
Ab initio cell wall invention, emergence, and evolution (2)
8 MIN READ
0
Allen Institute confirms food energy is information
6 MIN READ
0
RNA-mediated adult learning, memory, and neurogenesis
3 MIN READ
0
Energy-dependent allelic imbalances, viruses, and pathology
3 MIN READ
0
Ab initio cell wall invention, emergence, and evolution (1)
2 MIN READ
0
Biologically uninformed biologists fight back and lose
3 MIN READ
0
Theistic evolutionists fight back and lose (3)
4 MIN READ
0
Theistic evolutionists fight back and lose (2)
4 MIN READ
0
Theistic evolutionists fight back and lose
4 MIN READ
0
Young earth creationists refute theistic evolution
3 MIN READ
0
Charles Darwin refutes theistic evolution
2 MIN READ
0
Bill Gates refutes theistic evolution (prequel)
2 MIN READ
0
Bill Gates refutes theistic evolution (sequel)
8 MIN READ
0
Bill Gates refutes theistic evolution
4 MIN READ
0
Emily Witkin refutes theistic evolution
2 MIN READ
0
Pseudoscientists fail to refute theistic evolution
4 MIN READ
0
Stuart Kauffman refutes theistic evolution
3 MIN READ
0
May the anti-entropic force of sunlight be with you
4 MIN READ
0
Francis S. Collins refutes theistic evolution
6 MIN READ
0
Energy is information. Objections over ruled. You’re fired!
7 MIN READ
0
Happy Darwin Day (2017)
4 MIN READ
0
George Church refutes theistic evolution (3)
5 MIN READ
0
George Church refutes theistic evolution (2)
2 MIN READ
0
George Church refutes theistic evolution
5 MIN READ
0
Bacteriophages prove evolution is a lie
2 MIN READ
0
Combating evolution: Battlefield medicine vs politicized science (2)
3 MIN READ
0
Natural selection for codon optimality and quantum viruses
5 MIN READ
0
Physicists: Desperate Acts (revisited)
6 MIN READ
0
Energy as information and constrained endogenous RNA interference (3)
4 MIN READ
0
Energy as information and constrained endogenous RNA interference (2)
2 MIN READ
0
Dobzhansky 1973 and precision medicine (5)
3 MIN READ
0
Dobzhansky 1973 and precision medicine (4)
5 MIN READ
0
Dobzhansky 1973 and Precision Medicine (2)
5 MIN READ
0
Science journalists or paid propagandists? (4)
6 MIN READ
0
Dobzhansky 1973 and precision medicine
6 MIN READ
0
Twisted theories and weaponized facts
3 MIN READ
0
Autophagy: from pre-mRNAs to microRNAs, enhancers, QTLs et al.
6 MIN READ
0
Mutations: the “driving force” behind human brain complexity?
6 MIN READ
0
Mutations: the "driving force" behind human brain complexity?
6 MIN READ
0
Science journalists or paid propagandists? (3)
6 MIN READ
0
Science journalists or paid propagandists? (2)
3 MIN READ
0
Science journalists or paid propagandists?
5 MIN READ
0
Critical values expose virus-driven energy theft (2)
4 MIN READ
0
Theorists sell hidden energy (2)
4 MIN READ
0
Critical values expose virus-driven energy theft
6 MIN READ
0
Anti-entropic energy and the “Music of Life”
3 MIN READ
0
Anti-entropic energy and the "Music of Life"
3 MIN READ
0
Chirality is reality
6 MIN READ
0
Energy-dependent hydrogen bonds in supercoiled DNA
7 MIN READ
0
Energy-dependent chirality (2)
3 MIN READ
0
Energy-dependent chirality
6 MIN READ
0
Energy-dependent alternative splicings 1996 – 2016 (2)
5 MIN READ
0
Vietnam Veterans and others with glioblastoma
3 MIN READ
0
Energy-dependent sensory maps (1996-2016)
5 MIN READ
0
Energy-dependent alternative splicings 1996 – 2016
12 MIN READ
0
Energy-dependent oscillating gene networks organize life
4 MIN READ
0
2016 obfuscated facts about energy as information
5 MIN READ
0
More refutations of neo-Darwinian nonsense
7 MIN READ
0
Re-inventing mutation-driven evolution
3 MIN READ
0
Sudden death indel polymorphism
4 MIN READ
0
Anti-entropic virucidal energy as information
10 MIN READ
0
Bio-functional information
3 MIN READ
0
Bird-brained theorists
< 1 MIN READ
0
Energy-dependent self-organization and self repair
8 MIN READ
0
Expunging the distinguished public
2 MIN READ
0
Explorers who do not know what is known (2)
6 MIN READ
0
Explorers who do not know what is known
4 MIN READ
0
Energy-dependent natural translational selection
4 MIN READ
0
Autophagy is the antiphage defense strategy
3 MIN READ
0
Energy-dependent de novo creation and neurogenesis (2)
5 MIN READ
0
Energy-dependent de novo creation and neurogenesis
7 MIN READ
0
Theories vs facts about polycombic adaptation
3 MIN READ
0
Tasting light links energy from creation to adaptation
9 MIN READ
0
Politicized science: The demise of RNA-mediated.com?
3 MIN READ
0
Combating evolution: Battlefield medicine vs politicized science
6 MIN READ
0
The futility of The Battlefield FB group
10 MIN READ
0
Metabolic Phenotyping Research
2 MIN READ
0
Phylogenetic similarity sans energy
2 MIN READ
0
Happy biophysically constrained Thanksgiving (in the USA)
3 MIN READ
0
De novo gene creation: Ignoring the experimental evidence
7 MIN READ
0
Light, behavior and autophagy, a gender-specific risk factor
4 MIN READ
0
Controlled amino acid treatment of all pathology
4 MIN READ
0
Energy-dependent purifying selection / autophagy (2)
2 MIN READ
0
Energy-dependent purifying selection / autophagy (3)
2 MIN READ
0
Energy-dependent purifying selection / autophagy (4)
2 MIN READ
0
Energy-dependent purifying selection / autophagy (5)
2 MIN READ
0
Energy-dependent purifying selection / autophagy (6)
3 MIN READ
0
Energy-dependent purifying selection / autophagy
2 MIN READ
0
Optogenetics: Let there be virucidal light!
2 MIN READ
0
Base pairs, amino acids and phenotypes
10 MIN READ
0
Survivors of RNA-mediated terrorism
5 MIN READ
0
RNA-mediated terrorism
< 1 MIN READ
0
Building delicious base pairs in class. It’s elementary Watson and Crick
3 MIN READ
0
Building delicious base pairs in class. It's elementary Watson and Crick
3 MIN READ
0
RNA-mediated “repurposing” is autophagy
2 MIN READ
0
RNA-mediated "repurposing" is autophagy
2 MIN READ
0
Epigenetics and autophagy vs mutations and evolution (8)
4 MIN READ
0
Energy-dependent maternal-to-zygotic transition
6 MIN READ
1
Virus-driven mutation or amino acid substitution
5 MIN READ
0
Epigenetically effected energy-dependent fluorescence (2)
6 MIN READ
0
Epigenetically effected energy-dependent fluorescence
5 MIN READ
0
Epigenetics and autophagy vs mutations and evolution (7)
3 MIN READ
0
Epigenetics and autophagy vs mutations and evolution (5)
2 MIN READ
0
Epigenetics and autophagy vs mutations and evolution (4)
< 1 MIN READ
0
Epigenetics and autophagy vs mutations and evolution
< 1 MIN READ
0
From Precis to Proof in 6000 years (3)
2 MIN READ
0
From precis to pre-mRNA and proof of concept in F1, F2, F3
6 MIN READ
0
From Precis to Proof in 6000 years
9 MIN READ
0
Coulombic interactions facilitate polycombic adaptation
5 MIN READ
0
Top-down adaptation vs bottom-up evolution
6 MIN READ
0
Light ‘drives’ adaptation; nothing ‘drives’ evolution (3)
< 1 MIN READ
0
Light ‘drives’ adaptation; nothing ‘drives’ evolution (2)
3 MIN READ
0
Polycombic ecological adaptation as a science, not a theory (2)
12 MIN READ
0
Demoncrats fight polycombic ecological adaptation
4 MIN READ
0
Polycombic ecological adaptation as a science, not a theory
3 MIN READ
0
Biological evolution as a philosophy, not a science
6 MIN READ
0
Attacking Young Earth Creationists
5 MIN READ
0
Did evolution autophosphorylate your kinases? (3)
2 MIN READ
1
Predictable idiosyncratic evolved convergence
3 MIN READ
0
The natural success of RNAi and failed treatment
7 MIN READ
0
Pheromone-controlled autophagy
4 MIN READ
0
Virus-mediated hecatombic evolution
2 MIN READ
0
Hecatombic evolution via oncocers and oncohistones
9 MIN READ
0
Did evolution autophosphorylate your kinases? (2)
5 MIN READ
1
Did evolution autophosphorylate your kinases?
5 MIN READ
1
Nutrient-dependent autophagy
3 MIN READ
0
Nutrient-dependent pheromone-controlled autophagy (2)
6 MIN READ
0
Nutrient-dependent pheromone-controlled autophagy
3 MIN READ
0
Life is energy-dependent task management
3 MIN READ
0
Hypothesis free pseudoscience vs facts (5)
6 MIN READ
0
Hypothesis free pseudoscience vs facts (4)
5 MIN READ
0
Hypothesis free pseudoscience vs facts (3)
3 MIN READ
0
Hypothesis free pseudoscience vs facts (2)
5 MIN READ
0
Hypothesis free pseudoscience vs facts (1)
5 MIN READ
0
The last RNA-mediated theory killer
< 1 MIN READ
0
Base pairs, olfaction and RNA thermometers
4 MIN READ
0
Chromatin: The structure of DNA (2)
8 MIN READ
0
Chromatin: The structure of DNA (3)
4 MIN READ
0
Chromatin: The structure of DNA
6 MIN READ
0
Consciousness is simply food rearranged
2 MIN READ
0
Hydrogen-atom energy in DNA base pairs
6 MIN READ
0
A working model of “captured” genes
6 MIN READ
0
The Aquatic Ape / Waterside Ape divergence
6 MIN READ
0
Light energy-dependent active motifs
6 MIN READ
0
Light energy-induced base pair changes (2)
4 MIN READ
0
Light energy-induced base pair changes (1)
6 MIN READ
0
Displays of ecological adaptation (2)
3 MIN READ
0
DNA repair via junk DNA (1)
5 MIN READ
0
Displays of ecological adaptation
2 MIN READ
0
Unconstrained mutations, Zika virus, Kallmann’s syndrome
4 MIN READ
0
Linking RNA structure to function
3 MIN READ
0
How did the innate immune system evolve?
4 MIN READ
0
The Aquatic Ape: New evidence?
4 MIN READ
0
Plant microRNAs slow virus-driven aging
3 MIN READ
0
Protecting 4 billion people from virus-driven entropy
< 1 MIN READ
0
Increased soil pH and nutrient availability
4 MIN READ
0
Energy-dependent cellular communication
3 MIN READ
0
Conserved biophotonic emissions
7 MIN READ
2
Virus-driven energy theft: honeybee model
3 MIN READ
0
Antithetical conclusions (7)
3 MIN READ
0
Antithetical conclusions (5)
4 MIN READ
0
Antithetical conclusions (4)
3 MIN READ
0
Anthetical conclusions (2)
5 MIN READ
0
Antithetical conclusions (3)
3 MIN READ
0
Anthetical conclusions
3 MIN READ
0
Non-random pheromone-controlled cell type differentiation
3 MIN READ
0
RNAi: From magic bullet to billion dollar baby
4 MIN READ
0
Co-evolution and co-speciation replace neo-Darwinian nonsense
4 MIN READ
0
Energy-dependent natural fluorescence and bioluminescence
2 MIN READ
0
Pseudoscientists ignore what serious scientists prove
6 MIN READ
0
GC-rich neo-Darwinian train wreck
5 MIN READ
0
Biophotonics, glycobiology, quantized biodiversity (3)
< 1 MIN READ
0
Biophysically constrained cell type differentiation
3 MIN READ
0
From angstroms to ecosystems
7 MIN READ
0
Food supplement or licensed immunostimulant?
6 MIN READ
0
Do weak bosons evolve into leptons?
4 MIN READ
0
Q and A: Energy-dependent cell type differentiation
10 MIN READ
0
Biophotonics, glycobiology, quantized biodiversity (2)
19 MIN READ
0
The end of neo-Darwinism
5 MIN READ
0
The Origin of Information (2)
6 MIN READ
0
Did “Nature” kill Steve Jobs? (3)
10 MIN READ
0
Funding the Human Genome Project-Write
2 MIN READ
0
RNA-mediated programming of E.coli
< 1 MIN READ
0
Did “Nature” kill Steve Jobs? (2)
6 MIN READ
0
Did “Nature” kill Steve Jobs?
3 MIN READ
0
Virus-driven downsizing of the human brain (2)
< 1 MIN READ
0
Virus-driven downsizing of the human brain
2 MIN READ
0
MicroRNAs and sexual orientation
8 MIN READ
0
RNA methylation, learning, memory and behavior,
< 1 MIN READ
0
Innate immunity and multiplexing
7 MIN READ
0
Major transition ends use of silly theories
5 MIN READ
0
RNA methylation
2 MIN READ
0
Model vs theory: Progress report!
8 MIN READ
0
Masters of deception about nature
8 MIN READ
0
The Mind’s Eyes (revisited)
10 MIN READ
0
RNA methylation, behavior, and disease
6 MIN READ
0
Modelling life scientifically: RNA-mediated events
12 MIN READ
0
Energy-dependent RNA methylation (9)
2 MIN READ
0
Energy-dependent RNA methylation (8)
4 MIN READ
0
Energy-dependent RNA methylation (7)
7 MIN READ
0
Energy-dependent RNA methylation (6)
9 MIN READ
0
Energy-dependent RNA methylation (4)
5 MIN READ
0
Energy-dependent RNA methylation (3)
10 MIN READ
0
Energy-dependent RNA methylation (2)
5 MIN READ
0
Energy-dependent RNA methylation (1)
6 MIN READ
0
Secular humanism and ecoterrorism
4 MIN READ
0
Thermotolerance and longevity (2)
3 MIN READ
0
War Games: False Flag Terrorism
5 MIN READ
0
Energy-dependent biodiversity (4)
4 MIN READ
0
Energy-dependent biodiversity
5 MIN READ
0
The tipping point? 50,000 publications (3)
6 MIN READ
0
The tipping point? 50, 000 publications
4 MIN READ
0
Magic, Miracle, or Molecular Mechanism (3)
2 MIN READ
0
Countdown to Genetics and Genomics
< 1 MIN READ
0
Magic, Miracle, or Molecular Mechanism (2)
8 MIN READ
0
Energy-dependent creation and entropy
4 MIN READ
0
RNA-mediated physics, chemistry, and molecular epigenetics (5)
6 MIN READ
0
RNA-mediated physics, chemistry, and molecular epigenetics (4)
5 MIN READ
0
RNA-mediated physics, chemistry, and molecular epigenetics (3)
5 MIN READ
0
RNA-mediated physics, chemistry, and molecular epigenetics (2)
7 MIN READ
0
RNA-mediated physics, chemistry, and molecular epigenetics
9 MIN READ
0
Wasted Templeton Funding (4)
3 MIN READ
0
MicroRNAs and/or QTLs: Who buried what?
2 MIN READ
0
RNA splicing, genetic variation, and disease
2 MIN READ
0
Wasted Templeton Funding (3)
2 MIN READ
0
Wasted Templeton Funding (2)
2 MIN READ
0
Millions of water molecules vs years of evolution
2 MIN READ
0
Magic, Miracle, or Molecular Mechanism?
10 MIN READ
0
Molecular Diagnostics: What is unprotected life? (5)
5 MIN READ
0
Molecular Diagnostics: What is unprotected life (4)
2 MIN READ
0
Molecular Diagnostics: What is unprotected life (3)
5 MIN READ
0
Genome-gate: Follow the money?
4 MIN READ
0
Molecular Diagnostics: What is unprotected life? (2)
7 MIN READ
0
Molecular Diagnostics: What is unprotected life?
2 MIN READ
0
Virus-driven sex differences?
2 MIN READ
0
Ricki Lewis’ Time Machine (4)
8 MIN READ
0
Creating and/or programming the immune system
< 1 MIN READ
0
Half truths support theories without facts
4 MIN READ
0
YEC compared to Perry Marshall’s opinions
4 MIN READ
0
Ricki Lewis’ Time Machine (3)
5 MIN READ
0
Creating nutrient-dependent life with enough genes to survive
3 MIN READ
0
Confusing effects and affects of visual input
2 MIN READ
0
Selective reporting of inferences: examples of pseudoscience
14 MIN READ
0
Ignore the evidence: Rachel Feltman
5 MIN READ
0
Energy-dependent purpose vs teleophobic telorexia
12 MIN READ
0
Stress-linked population-level history dependence
5 MIN READ
0
Jay R. Feierman: twenty years of antagonism
5 MIN READ
0
Do not miss the misrepresentations
2 MIN READ
0
Energy dependent RNA-mediated immunity (5)
4 MIN READ
0
Energy dependent RNA-mediated immunity (4)
6 MIN READ
0
Energy dependent RNA-mediated immunity (3)
8 MIN READ
0
Energy dependent RNA-mediated immunity (2)
10 MIN READ
0
Energy-dependent RNA-mediated immunity (1)
10 MIN READ
0
RNA-mediated DNA modifications
5 MIN READ
0
The toxic river of neo-Darwinian pseudoscience
3 MIN READ
0
Science vs semantics
4 MIN READ
0
From angstroms to ecosystems and entropy
2 MIN READ
0
Bacteria see the light and they adapt (2)
5 MIN READ
0
Soil bacteria, bulls, cows, microRNAs, and mammary glands (2)
3 MIN READ
0
Soil bacteria, bulls, cows, microRNAs, and mammary glands
3 MIN READ
0
Bringing RNA back to epigenetics (20 years later)
11 MIN READ
0
Effects on invertebrate GnRH and affects on primate behavior
6 MIN READ
0
Hybrids: making microbes and Democrats into monkey’s uncles?
11 MIN READ
0
Creating gravity, nucleic acids, receptors, and supercoiled DNA (2)
9 MIN READ
0
Nutrient-dependent RNA-mediated cause and effect
3 MIN READ
0
Creating gravity, nucleic acids, receptors, and supercoiled DNA
2 MIN READ
0
Hydrogen-atom transfer in DNA base pairs (7)
5 MIN READ
0
Bacteria see the light and they adapt
7 MIN READ
0
The Light and Darkness of “Evolution 2.0”
11 MIN READ
0
Will modern human populations adapt to the Zika virus?
5 MIN READ
0
Models of scientific literacy
3 MIN READ
0
Organic Compounds and the Miracle of Smell and Taste
7 MIN READ
0
Neuroscience Virtual Event vs AAAS Symposium
3 MIN READ
0
Cancer: Evolution 2.0’s Blind Spot
13 MIN READ
0
Despicable fools?
4 MIN READ
0
Ricki Lewis’ Time Machine
5 MIN READ
0
Virus-perturbed alternative splicings
3 MIN READ
0
Hydrogen-atom transfer in DNA base pairs (5)
10 MIN READ
0
The radical pair mechanism of ecological speciation
3 MIN READ
0
Brain evolution?
< 1 MIN READ
0
Origin and evolution of the genetic code
2 MIN READ
0
Center stage RNA-mediated events (since 1996)
3 MIN READ
0
The unoffically uninvited
3 MIN READ
0
Hydrogen-Atom Transfer in DNA Base Pairs (4)
4 MIN READ
0
Blood test links atoms to ecosystems
2 MIN READ
0
Another failed rescue attempt
5 MIN READ
0
Juvenile hormone links atoms to ecosystems
3 MIN READ
0
Stress-induced energy-dependent DNA repair
4 MIN READ
0
A failed theory of cancer: two more decades of pseudoscience
5 MIN READ
0
Teleophobes vs teleophiles: a recent history
2 MIN READ
0
Ecological genomics: teleophobes respond (too late)
10 MIN READ
0
Intelligent RNAs vs evolved genes
2 MIN READ
0
Genes, orchid odors, and pheromones from blonds
5 MIN READ
0
Amino acid substitutions, stress, and human behavior
3 MIN READ
0
RNA-mediated theory killers (11) (12) (13) (14) (15)
7 MIN READ
0
Finding peace and π in the light of H bond energy (2)
8 MIN READ
0
MicroRNA-mediated RNA epigenetics
2 MIN READ
0
RNA methylation, RNA-directed DNA methylation, learning and memory
3 MIN READ
0
Finding peace and π in the light of H bond energy
5 MIN READ
0
Epigenetic (re)programming of behavior (3)
5 MIN READ
0
Perry Marshall: too much information for atheist PZ Myers
3 MIN READ
0
Epigenetic (re)programming of behavior (2)
6 MIN READ
0
Assumptions prove ignorance (2)
5 MIN READ
0
Epigenetic (re)programming of behavior
4 MIN READ
0
Assumptions prove ignorance
5 MIN READ
0
Neo-Darwinian sink testing
2 MIN READ
0
NORAD: air defense and defense against genomic entropy
7 MIN READ
0
Life history transitions and RNA-mediated survial
2 MIN READ
0
Did Brain Atrophy Evolve?
2 MIN READ
0
Did Dobzhansky see the UV light of creation?
10 MIN READ
0
Researchers rename, reshuffle, and reveal their ignorance
3 MIN READ
0
RNA-mediated theory killers (6) (7) (8) (9) (10)
5 MIN READ
0
Does metabolism link beneficial mutations to cancer?
10 MIN READ
0
Hydrogen-Atom Transfer in DNA Base Pairs (3)
3 MIN READ
0
Manufacturing fossil “evidence”
4 MIN READ
0
RNA-mediated theory killers (4)
9 MIN READ
0
RNA-mediated theory killers (5)
2 MIN READ
0
RNA-mediated theory killers (3)
7 MIN READ
0
Ecological adaptations you cannot live without
6 MIN READ
0
RNA-mediated regulatory mechanisms link microbes to humans (3)
5 MIN READ
0
Essential pseudoscientific concepts of atheism
3 MIN READ
0
RNA-mediated theory killers (2)
8 MIN READ
0
RNA-mediated theory killers
6 MIN READ
0
Natural cooperation and Evolution 2.0
6 MIN READ
0
A million dollar paradox?
2 MIN READ
0
A two-faced protein enables RNA-mediated DNA repair (4)
6 MIN READ
0
A two-faced protein enables RNA-mediated DNA repair (5)
4 MIN READ
0
A two-faced protein enables RNA-mediated DNA repair (3)
8 MIN READ
0
Teaching the biologically uninformed
3 MIN READ
0
MicroRNAs vs Red Queen hypothesis
4 MIN READ
0
Innovative Neurotechnologies Link Sunlight to Precision Medicine
4 MIN READ
0
Hydrogen-Atom Transfer in DNA Base Pairs
9 MIN READ
0
Sulfated steroids and RNA-mediated identity codes
< 1 MIN READ
0
Neo-Darwinism vs the neocortex
4 MIN READ
0
A two-faced protein enables RNA-mediated DNA repair
2 MIN READ
0
Life and death predicted by DNA methylation
3 MIN READ
0
Nutrient-dependent trophic analogs
3 MIN READ
0
Phenols: Laws of physics meet biology
2 MIN READ
0
Plasma created by sunlight and RNA–mediated epigenetic heredity
2 MIN READ
0
Positive feedback loops and epigenetic traps
2 MIN READ
0
Stress-perturbed mitochondrial dysfunction (2)
5 MIN READ
0
Behavioral Immune System model
4 MIN READ
0
Stress-perturbed mitochondrial dysfunction
4 MIN READ
0
Understanding cell type differentiation
3 MIN READ
0
Let there be anti-entropic light (3)
6 MIN READ
0
Theorists can’t understand biology
6 MIN READ
0
Neuroplasticity
4 MIN READ
0
Sensationalizing no new mechanism
2 MIN READ
0
Cell type differentiation: atoms to ecosystems
2 MIN READ
0
Too complex for the Complex Biological Systems Alliance
4 MIN READ
0
Receptor methylation controls behavior
2 MIN READ
0
Rediscovering quantum behavior
2 MIN READ
0
From dust to genomic entropy?
3 MIN READ
0
Virus, transposon and plasmid evolution
< 1 MIN READ
0
Models are not theories (2)
2 MIN READ
0
Models are not theories (1)
2 MIN READ
0
MicroRNA-encoded behavior
7 MIN READ
0
Fossils vs cell types and the brain
3 MIN READ
0
Can veterans and other prisoners escape pseudoscience?
2 MIN READ
0
eQTLs and ecological adaptation
< 1 MIN READ
0
Unraveling the secrets of RNA-mediated events
2 MIN READ
0
RNA central and RNA-mediated.com
6 MIN READ
0
Ecological speciation. Get it, theorists?
< 1 MIN READ
0
Designing, engineering, and protecting biodiversity
3 MIN READ
0
The virome, microbiome, replisome and supercoiled DNA
2 MIN READ
0
Skip the politics; embrace the facts
5 MIN READ
0
Controled Stem Cell Expansion
3 MIN READ
0
Neo-Darwinian logic is nonsense
3 MIN READ
0
Human pheromone deniers: What’s next?
5 MIN READ
0
FREE* SAMPLE: Histone modification
4 MIN READ
0
700 million years of evolution?
5 MIN READ
0
Biophysically constrained or unconstrained?
2 MIN READ
0
Mutated mitochondrial genes vs Supercoiled DNA
2 MIN READ
0
RNA-mediated retrotransposon-mediated biodiversity
3 MIN READ
0
Mystery machine vs model (3)
2 MIN READ
0
Mystery machine vs model (2)
2 MIN READ
0
Mystery machine vs model
2 MIN READ
0
Mystery machine vs medical intelligence
2 MIN READ
0
Epigenetic inheritance of stress-perturbed protein folding
2 MIN READ
0
Preventing genomic entropy
4 MIN READ
0
The Current State of Neuroscience
3 MIN READ
0
Cancer: forward and reverse
2 MIN READ
0
Energy and evolution: another opinion?
2 MIN READ
0
Keep the gay gene theory?
< 1 MIN READ
0
Bird watchers and RNAs in cancers
2 MIN READ
0
Supercoiled DNA constrains virus-driven genomic entropy
2 MIN READ
0
Let there be anti-entropic light (2)
4 MIN READ
0
Foundamentals of theory
4 MIN READ
0
Microbes to humans 2015 Nobel Prize
2 MIN READ
0
Non-mainstream scientist shares Nobel prize in Medicine
3 MIN READ
0
Is mainstream science in “Science” pseudoscience?
5 MIN READ
0
Mechanisms of stress: from genes to cancer
9 MIN READ
0
Viruses come alive: Tree of life pseudoscience
6 MIN READ
0
Theorists have not seen the light (2)
3 MIN READ
0
Gene creation (revisited)
2 MIN READ
0
Hijacked light energy and vertebrate pathology
4 MIN READ
0
Theorists have not seen the light
4 MIN READ
0
The creation and activation of GPCRs
4 MIN READ
0
A Darwinian pediatrician and expected life span
2 MIN READ
0
A 5-10K comparison of design principles to evolution
3 MIN READ
0
Alternative pre-mRNA splicing and ecological adaptation
7 MIN READ
0
Pathology: Eating and breathing viruses
2 MIN READ
0
Nucleic acids: Stability of DNA/RNA
2 MIN READ
0
Exosomes and the RNA-mediated future of medicine (3)
2 MIN READ
0
Exosomes and the RNA-mediated future of medicine (2)
< 1 MIN READ
0
Exosomes and the RNA-mediated future of medicine
5 MIN READ
0
Multi-omic analysis features (SNPs, miRNA)
3 MIN READ
0
Anti-entropic effects on the origin of life
< 1 MIN READ
0
Somatic hypermutation vs RNA-mediated events
2 MIN READ
0
From fertilization to RNA-mediated events and back
2 MIN READ
0
Phytochemical link from the sun to cell types
2 MIN READ
0
Metabolic competition and cancer
2 MIN READ
0
Natural selection: an anti-entropic force?
2 MIN READ
0
Genome sequencing, cadherins, and quantum consciousness
6 MIN READ
0
“New” quantum biology. Pirating the old
3 MIN READ
0
Creating genes and species
3 MIN READ
0
Cell types, SNVs, CNVs, and chromosomes
3 MIN READ
0
A “new” code enables ecological adaptation
4 MIN READ
0
Thermotolerance and Longevity
57 MIN READ
0
Is life the balance between quantum and classical physics?
3 MIN READ
0
Anti-entropic containment of energy: symbiosis 1.0
5 MIN READ
0
RNA-mediated X chromosome inactivation
3 MIN READ
0
The “great filter” is an epigenetic trap
4 MIN READ
0
Atomic-resolution of cell type signaling
4 MIN READ
0
Information and communication (2)
6 MIN READ
0
Information and communication
2 MIN READ
0
Unraveling evolutionary pseudoscience
2 MIN READ
0
Eibi Nevo gets it wrong
3 MIN READ
0
Rs3827760 is Val370Ala and EDARV370A
3 MIN READ
0
From “Blood Music” to Evolution 2.0
5 MIN READ
0
Olfaction & the octopus and human genomes (2)
4 MIN READ
0
Olfaction & the octopus and human genomes
3 MIN READ
0
Thermodynamic constraints did not evolve
3 MIN READ
0
Protosuns, prebiotic molecules, proteins, and people
4 MIN READ
0
Mammalian-wide interspersed repeats (MIRs)
2 MIN READ
0
Ecological speciation vs revised evolutionary syntheses
6 MIN READ
0
Ecology replaces the extended evolutionary synthesis
4 MIN READ
0
The stability of organized genomes (4)
3 MIN READ
0
The stability of organized genomes (3)
3 MIN READ
0
The stability of organized genomes (2)
5 MIN READ
0
Epimutation.com: a domain of confusion
8 MIN READ
0
Light -induced nucleic acid-mediated gene duplication?
< 1 MIN READ
0
Is the best Chinese research from China?
3 MIN READ
0
The stability of organized genomes
5 MIN READ
0
Viruses, amino acids, and somatic cell types (3)
9 MIN READ
0
Viruses, amino acids, and somatic cell types (2)
5 MIN READ
0
Viruses, amino acids, and somatic cell types
4 MIN READ
0
Big Bang Cosmology vs Reality
< 1 MIN READ
0
RNA-mediated terms of virus-induced en-deer-ment
2 MIN READ
0
Domestication via a single amino acid substitution
2 MIN READ
0
Hematopoiesis and practopoiesis
4 MIN READ
0
Koonin: viruses killed neo-Darwinism
3 MIN READ
0
RNA-mediated morphological AND behavioral phenotypes
2 MIN READ
0
Becoming biologically informed (3)
3 MIN READ
0
Easy editing: Reinventing our RNA world
3 MIN READ
0
Is photic-zone ribosomal diversity linked to all biodiversity?
2 MIN READ
0
Picornaviruses moving between primates (or not)
2 MIN READ
0
Riding the wrong direction
3 MIN READ
0
Decreased phenotypic variation: Faith in Facts
7 MIN READ
1
Faithfully repaired DNA
5 MIN READ
0
RNA-mediated gene duplication, fixation, and ecological adaptation
6 MIN READ
0
MicroRNA controlled growth and brain development
7 MIN READ
0
MicroRNA – controlled ecological adaptations
5 MIN READ
0
Becoming biologically informed
2 MIN READ
0
The RNA-mediated sum of our parts
2 MIN READ
0
Celebrating independence from ridiculous theories
3 MIN READ
0
“New” epigenetic mechanism for lifelong learning?
3 MIN READ
0
The sum of our RNA-mediated parts
2 MIN READ
0
microRNAs and memory: Why trust a theory?
2 MIN READ
0
Protein folding and Google page rank
2 MIN READ
0
Unconscious affect (revisited)
2 MIN READ
0
Iron, ferritin, thyroxine
2 MIN READ
0
Virus-driven death by evolutionary theory
3 MIN READ
0
“Evolution” of sex differences?
2 MIN READ
0
Olfactory perception of genetic information
2 MIN READ
2
Nutrient stress-induced RNA-mediated pathology
< 1 MIN READ
0
Amino acid substitutions are not mutations
2 MIN READ
0
RNA-mediated events from A to Z
2 MIN READ
0
What I cannot create I eliminate from discussion
3 MIN READ
0
RNA-mediated development (3)
5 MIN READ
0
RNA-mediated development (2)
< 1 MIN READ
0
RNA-mediated development
2 MIN READ
0
Creating and maintaining the human virome
2 MIN READ
0
Healthy mutants
2 MIN READ
0
Uniquely epigenomic gene regulation
2 MIN READ
0
MicroRNAs and inappropriate functions
2 MIN READ
0
Rejecting pseudoscientific nonsense
6 MIN READ
1
Living the life that randomness created? (Sarcasm alert)
3 MIN READ
0
Ignoring top-down causation
2 MIN READ
0
30 years of theoretical nonsense
3 MIN READ
0
microRNAs, glycosylation, and genomes
4 MIN READ
0
Unknown mechanisms and conclusions
2 MIN READ
0
Alternative splicings: epigenetics meets pharmacogenomics
4 MIN READ
0
Physicians who practice evolutionary medicine?
3 MIN READ
0
Pattern recognition: biogeochemical structure and function
5 MIN READ
0
Viruses and the human-like microbiome
3 MIN READ
0
Informing the biologically uninformed
< 1 MIN READ
0
Tweaking the story to fit the theory
5 MIN READ
0
Appetite for ingesting theories (raw)
< 1 MIN READ
0
I forgot. How do mutations cause evolution?
4 MIN READ
0
One amino acid substitution, genes, and brain activity
3 MIN READ
0
Batch effect vs epigenetic effects
3 MIN READ
0
Gene expression, immortality, and cancer
6 MIN READ
0
Missing a fact: microRNAs are genomic biomarkers
4 MIN READ
1
Bee-birthed epigenetics and primate cell types
5 MIN READ
0
A lighting requirement for life
11 MIN READ
3
Scientists lose. A sci-fi author gains credibility
2 MIN READ
0
Vitamin B3 and DNA repair
4 MIN READ
0
Bees and primates automagically evolve
3 MIN READ
0
Ignoring systems complexity (it’s too complicated)
3 MIN READ
0
Five years of Ferguson
2 MIN READ
0
Computing via phosphorylation and fixation
3 MIN READ
0
Virus-driven cancer treatment and prevention
2 MIN READ
0
The microbiome, pharmacogenetics, and privacy
< 1 MIN READ
0
A special issue on nutritional epigenetics
5 MIN READ
0
Retinoic acid + one receptor regulate the genome
3 MIN READ
0
Epigenetics: microRNAs effect an integrative pathway
4 MIN READ
0
Protein isoforms do not evolve
3 MIN READ
0
Tissue type variation and expression of genes
< 1 MIN READ
0
From gut bacteria to breast milk and back
5 MIN READ
0
Chance mutations — not natural selection
5 MIN READ
0
Viruses in gut microbes
4 MIN READ
0
How to find a “holy grail” under your nose
3 MIN READ
0
Physics, chemistry, light, and life
2 MIN READ
0
MicroRNAs and the exposome
5 MIN READ
0
Finding odor and taste receptors everywhere
6 MIN READ
0
Linking the origin of birds to dinosaurs
< 1 MIN READ
0
Epigenetics vs the fossil record
4 MIN READ
0
An epigenetic mark links algae, worms, and flies
2 MIN READ
0
Effect and affect of a single base-pair change
4 MIN READ
0
Tet3 regulation of nutrient-dependent cell type differentiation
4 MIN READ
0
Thermodynamics and protein folding landscapes
2 MIN READ
0
RNA-mediated silencing of a chromosome
3 MIN READ
0
Viruses, proteins, and gut metagenomes do not evolve
< 1 MIN READ
0
Pathology constrains X-linked evolution
2 MIN READ
0
Re-inventing a completely new thing
4 MIN READ
0
Feedback loops link insects to human brains
2 MIN READ
0
Viruses and cell type differentiation
3 MIN READ
0
RNA-mediated cell types and precision medicine
5 MIN READ
0
DNA Methylation and organized genomes (2)
10 MIN READ
0
DNA Methylation and organized genomes
7 MIN READ
0
Misunderstanding cancer
7 MIN READ
0
Amino acid-dependent cell type differentiation
8 MIN READ
0
Nutrient-dependent RNA interference (2)
4 MIN READ
0
The Darwin Code by Greg Bear
13 MIN READ
0
Nutrient-dependent RNA interference
4 MIN READ
0
Mutisensory integration: watching the paradigm shift
12 MIN READ
0
Origin of life and cancer (1,2,3)
2 MIN READ
0
A genetic variant refutes neo-Darwinism
6 MIN READ
0
MicroRNAs and invasive phenotypes
< 1 MIN READ
0
Silencing genes and serious scientists
3 MIN READ
0
The miRNA/mRNA balance: a suboptimal strategy?
2 MIN READ
0
Nutrient-dependent microRNAs control cell types
6 MIN READ
0
Life: conserved ion and amino acid transporters
3 MIN READ
0
Deeply ingrained thoughts
< 1 MIN READ
0
Graphic misrepresentations of ecological adaptation
2 MIN READ
0
Too many targets for theories
3 MIN READ
0
Methylation maintains cell type differences (2)
2 MIN READ
0
Methylation maintains cell type differences
3 MIN READ
0
Creating nothing but a theory (3)
4 MIN READ
3
Creating nothing but a theory (2)
4 MIN READ
0
Creating nothing but a theory
4 MIN READ
0
Viruses and ecologically adapted animals
2 MIN READ
0
2 genes in 2 species (too expensive and too insignificant)
11 MIN READ
0
The quantum / classical RNA-mediated ‘tipping point’
3 MIN READ
0
Epigenetic effects of soil bacteria on plants
< 1 MIN READ
0
Epigenetic switch links MicroRNAs to RNA-protein interactions
4 MIN READ
0
Quantum correlations/pseudoscience
3 MIN READ
0
Anti-entropic solar energy
5 MIN READ
0
Mimicking claims and ignoring facts
4 MIN READ
0
Correctly modeling life on this planet
3 MIN READ
0
Correctly modeling biological energy
2 MIN READ
0
Correctly modeling ecological adaptation
4 MIN READ
0
Behavior (4): All responses are RNA-mediated in birds
4 MIN READ
0
Questions about life’s diversity
21 MIN READ
0
UV-light mutations and gene loss (not gain)
5 MIN READ
0
Virus-driven origin of life
2 MIN READ
0
Luis P. Villarreal tells it like it is
3 MIN READ
0
RNA-directed gene choice
5 MIN READ
0
Walk towards the light
< 1 MIN READ
0
Two types of microRNA are not double agents
2 MIN READ
0
Epigenetics Skeptism
5 MIN READ
0
Is DNA-directed transcription RNA-mediated?
2 MIN READ
0
Assembling yourself: Molecular self / other recognition
4 MIN READ
0
What about birds?
5 MIN READ
0
Virus-driven cell type differentiation
2 MIN READ
0
RNA directed DNA methylation and cell types
4 MIN READ
0
Implicating microRNAs in cancer
2 MIN READ
0
Rejecting what is known about viral microRNAs and nutrient-dependent microRNAs
2 MIN READ
0
Reverse phosphorylation
7 MIN READ
0
RNA-mediated repurposing in microbes and adaptations in primate brains
2 MIN READ
0
RNA-mediated “repurposing” is nutrient-dependent and pheromone-controlled
3 MIN READ
0
RNA-mediated "repurposing" is nutrient-dependent and pheromone-controlled
3 MIN READ
0
From 3-D to epigenetically-effected 4-D genome make-up
5 MIN READ
0
Let there be anti-entropic light (1)
11 MIN READ
0
An epigenetic trap (the prequel)
5 MIN READ
0
Imagining that data historically supports evolutionary theory
7 MIN READ
0
Atheism: Arrogant, useless, and divisive ignorance
7 MIN READ
0
The anti-entropic force of "Nature"
2 MIN READ
0
Nutritional epigenetics, exercise, and immune system integrity
4 MIN READ
0
What if Darwin was not still dead?
2 MIN READ
0
Military combat training to fight disease (2)
3 MIN READ
0
Military combat training to fight disease
3 MIN READ
0
Theoretical physics and molecular biology
5 MIN READ
0
Atoms to ecosystems is not almost a molecular ecology
3 MIN READ
0
Epigenetic effects of viruses on cellular homeostasis (2)
2 MIN READ
0
RNA-mediated epigenetic modification via DNA-methylation
4 MIN READ
0
Quantum physics, quantum biology, and quantum consciousness
4 MIN READ
0
How fast can evolutionary theory be changed?
5 MIN READ
0
Biological energy and a microbiome model of a light-driven time machine
3 MIN READ
0
The biologically-based origin of the mammalian placenta (2)
6 MIN READ
0
Quantum Superpositions: let there be light
4 MIN READ
0
Are viruses microRNAs? (2)
5 MIN READ
0
Are viruses microRNAs?
3 MIN READ
0
Sneaking up from behind (2)
3 MIN READ
0
Sneaking up from behind
4 MIN READ
0
Quantum entanglement, mass, and biomass
4 MIN READ
0
Physicists: Desperate Acts
16 MIN READ
0
An epigenetic trap (the sequel)
10 MIN READ
0
Mute points: most are afraid to mention them
6 MIN READ
0
All of “like kind” (Part 2)
2 MIN READ
0
All of "like kind" in the (bigger) family
6 MIN READ
1
Environment epigenetically shapes the immune system
3 MIN READ
0
Beneficial microbes kill beneficial mutations
4 MIN READ
0
Constrained evolution is ecological adaptation
5 MIN READ
0
A single amino acid substitution differentiates cell types of E. coli
3 MIN READ
0
Unconstrained evolutionary innovability
4 MIN READ
0
RNA-protein interactions reveal biophysical to ecological landscapes
2 MIN READ
0
Mutagenesis: Replacing facts with theories
5 MIN READ
0
ISHE's human ethology group
2 MIN READ
0
Amino acid homeostasis for a Happy New Year!
6 MIN READ
0
Understanding cell type differentiation
3 MIN READ
0
From Hydra to humans vs a Lakatosian research program
4 MIN READ
0
Unified nutritional and molecular mechanisms
9 MIN READ
0
Removing natural selection; reshaping the horse; adjusting evolutionary theory
4 MIN READ
0
Communication, not mutations
2 MIN READ
0
Models by evolutionary biologists are not models
2 MIN READ
0
The future of physics predicts no future for evolutionary theory
3 MIN READ
0
Model organisms: the birds and the bees
3 MIN READ
0
Chemical ecology and RNA-mediated control of DNA loops
< 1 MIN READ
0
Atoms to ecosystems: Evolutionary theory vs the coelacanth
6 MIN READ
0
Jumping back: Science or Pseudoscience? (2)
2 MIN READ
0
Jumping back: Science or Pseudoscience?
6 MIN READ
0
Single-cell level assay of protein biosynthesis and degradation
2 MIN READ
0
Glycine and GnRH: Am I being pedantic?
4 MIN READ
0
Extensive molecular evidence vs ridiculous theories
2 MIN READ
0
Dual genomes: exposing the evolution industry
5 MIN READ
0
One test of bioenergetic health?
2 MIN READ
0
Meaningful dialogue, anonymous fools and idiot minions
4 MIN READ
0
Epigenetic pharmacology and RNA-mediated transciptional landscapes
3 MIN READ
0
RNA-mediated events and "The Theory of Everything"
4 MIN READ
0
Sackler Colloquium: Effects or AFFECTS on Behavior
2 MIN READ
0
Thermodynamic constraints and ecological adaptations sans evolution
6 MIN READ
0
Eliminating correlations from evolutionary ecology
6 MIN READ
0
RNA-mediated species specificity
2 MIN READ
0
Are mutations beneficial?
4 MIN READ
0
The key to science: experimental evidence
5 MIN READ
0
Ecological adaptations reported as evolution in insects and mammals
4 MIN READ
0
There’s a model for that!
5 MIN READ
0
Biologists puzzled by evolved RNAs and decaying DNA
6 MIN READ
0
SNPs and millions of small variations in the human genome
3 MIN READ
0
More than a bag of chemicals?
2 MIN READ
0
Achiral GnRH: no prophesy, just prediction
9 MIN READ
0
Nothing new under the sun, except pseudoscientific nonsense
6 MIN READ
0
Making sense of quotes scattered across disciplines
2 MIN READ
0
Intelligent viruses and cancers?
10 MIN READ
0
Complex behaviors of cell types in cancer
6 MIN READ
0
De novo DNA methylation?
3 MIN READ
0
RNA eclipses the importance of DNA to cell type differentiation
4 MIN READ
0
We need pattern recognition, not proclamations
3 MIN READ
0
We need pattern recognition, not a prologue
6 MIN READ
0
Pharmacogenomics
3 MIN READ
0
Sexual differentiation of cell types in plants
2 MIN READ
0
A model of MHC 'evolution'
2 MIN READ
0
From deep time into real time: What evolutionary processes?
10 MIN READ
0
In theory, or supported by experimental evidence?
5 MIN READ
0
It’s cell type differention, not cell fate determination (2)
3 MIN READ
0
Are evolutionary theorists 'nob ends'?
5 MIN READ
2
ALPHA GENOMIX
7 MIN READ
2
No excuses: Creation and the meaning of organismal complexity
7 MIN READ
0
Nutrient-dependent gene duplication in plants (but not animals?)
4 MIN READ
0
Evolutionary theorists justify fear of the Ebola viruses
2 MIN READ
0
Evolutionary theorists and evolutionary theists live under rocks
< 1 MIN READ
0
No understanding of biodiversity
2 MIN READ
0
Understanding physical forces of ecological variation and adaptation
< 1 MIN READ
0
Eliminating evolutionary theory
3 MIN READ
0
Behavioral ecology: please continue to believe in our fantasies
3 MIN READ
0
2014 and 2004 Nobel Prize in Medicine
3 MIN READ
0
RNA-mediated events: chromosomal rearrangements and genomic rearrangements
2 MIN READ
2
How much good can be attributed to social science theories?
< 1 MIN READ
0
RNA-mediated genetic engineering (Part 3)
< 1 MIN READ
0
RNA-mediated genetic engineering (Part 2)
3 MIN READ
0
RNA-mediated genetic engineering
2 MIN READ
0
RNA-mediated events are not a matter of faith
2 MIN READ
0
Color vision refutes the evolutionary dogma of gene duplication
4 MIN READ
0
Unassailable evidence vs assumptions
4 MIN READ
0
Forces of "Nature" limit dissemination of information
4 MIN READ
0
Genomic surveillance ends our world of RNA-mediated ecological adaptations
2 MIN READ
0
Systems biology and memory disorders
3 MIN READ
0
RNA-mediated cell type differentiation and behavior
3 MIN READ
0
Different physical locations and different molecular mechanisms of health and disease
2 MIN READ
0
Metabolism, fixation, health or neurodegerative disorder
2 MIN READ
0
Physics, Chemistry, and Molecular biology (PCMb)
2 MIN READ
0
Physics denied; pseudoscientific nonsense accepted
2 MIN READ
0
Ecological variation and niche construction: 1, 2, 3
6 MIN READ
0
Epigenetically-effected metabolic shifts and ecological adaptations
3 MIN READ
0
Evolving DNA before RNA
4 MIN READ
0
Stop evolutionary theorists. Kill cancers
3 MIN READ
0
Did our adapted mind evolve? (Revisited)
2 MIN READ
0
De novo gene Creation sans evolution of genes via mutations
4 MIN READ
0
Did our adapted mind evolve?
6 MIN READ
0
RNA-directed DNA methylation and RNA-mediated events
5 MIN READ
1
Miracles are not miracles to evolutionary theorists
2 MIN READ
0
Mathematical model: microRNA and epigenetic regulation
2 MIN READ
0
Seemingly futile cycles are not thermodynamically futile
3 MIN READ
0
Do bacterial proteins evolve?
3 MIN READ
0
“Kardashians” in science
< 1 MIN READ
0
Probable changes in connectivity
2 MIN READ
0
New technique used in report of atomic-level ecological adaptations
3 MIN READ
0
Can epigenetic inheritance occur without concurrent changes in morphology AND behavior?
3 MIN READ
0
Memory of repression and memory of behavior (2)
< 1 MIN READ
0
Memory of repression and memory of behavior
2 MIN READ
0
Quantum physics meets Evolutionary Psychology News
2 MIN READ
0
Nutrient-dependent pheromone-controlled exercise-induced physiques
< 1 MIN READ
0
Watching and waiting for more retractions
2 MIN READ
0
RNA-mediated ecological adaptations of teeth
4 MIN READ
0
A molecular visualizer of worthwhile molecular biology
2 MIN READ
0
RNA-mediated species diversification from microbes to primates
3 MIN READ
0
RNA-mediated events found everywhere
2 MIN READ
0
Reseachers think copy number variation is genetically determined
< 1 MIN READ
0
Some neuroscientists think a mutation led to human language development
< 1 MIN READ
0
Genotype, observed phenotype, and distinctly disordered behaviors
3 MIN READ
0
Is the problem an internet echo or Feierman's ethics?
2 MIN READ
0
Behavior (3): All responses are RNA-mediated in bees
3 MIN READ
0
RNA-mediated ecological adaptation is not evolution
2 MIN READ
0
Exploding genomes and chromosomal rearrangements via RNA-mediated events
2 MIN READ
0
Behavior (2): All responses are RNA-mediated not genetically-determined
4 MIN READ
0
Behavior: The first response is RNA-mediated not genetically-determined
3 MIN READ
0
Mechanisms that are not understood increase clarity
4 MIN READ
2
Pattern recognition and conserved receptors (TAARs)
5 MIN READ
0
Nutrient-dependent erythropoiesis and anemia
6 MIN READ
0
Evolutionary heritage or ecological adaptation? Racism versus reality
4 MIN READ
0
A relatively young branch of science called epigenetics
4 MIN READ
0
Insect homology and diversity attributed to mutations
2 MIN READ
0
Biophysically constrained beginnings of RNA and DNA
4 MIN READ
0
Baby talk: More misrepresentations of ecological adaptations
2 MIN READ
0
Comparing divergent model organisms
2 MIN READ
1
A microRNA-mediated mechanism that is epigenetically inherited
2 MIN READ
0
Epimutations: Attacking pseudoscientific dogmas
4 MIN READ
0
Order and disorder: Ecological adaptations not mutations
6 MIN READ
0
microRNAs differentiate neuronal cell types
3 MIN READ
0
microRNAs and species relationships
3 MIN READ
0
Drunks and Monkeys: Pseudoscientific nonsense
2 MIN READ
0
The quantum biology of consciousness
3 MIN READ
0
Behavior is receptor-mediated
10 MIN READ
0
Genes and Race: Human History?
2 MIN READ
0
Soft atheism: a case for Creation
< 1 MIN READ
0
Nutrition, pheromones and cancer (2)
2 MIN READ
0
Ecologically linked adapted ants and brains
2 MIN READ
0
MiRNAs methylation and ecological adaptation sans mutations
2 MIN READ
0
Not focused and self-aggrandizing
2 MIN READ
0
Nutrient-dependent cooperation vs cannibalism (video)
2 MIN READ
0
Cell-type differentiation
2 MIN READ
0
Another powerful refutation of mutation-initiated natural selection
4 MIN READ
0
The new light of chromosomal rearrangements
3 MIN READ
0
Darwinian theories vs Darwin's facts
2 MIN READ
0
Chromosomal rearrangements and ecological adaptations
4 MIN READ
0
The wrong picture of our evolution
4 MIN READ
0
96 fixed amino acid substitutions, not 96 genes
6 MIN READ
0
Jay R. Feierman
3 MIN READ
0
Don't tell the Creationists
2 MIN READ
0
Targeting a cancer gene
2 MIN READ
0
Alternative splicings (very technical)
2 MIN READ
0
Pulses of olfactory/pheromonal input
2 MIN READ
0
Social experiences epigenetically effect gene networks
2 MIN READ
0
Pheromone-controlled thermodynamics and cancer
2 MIN READ
0
Seeds of life: astrobiological theory and mutations theory
3 MIN READ
0
Your comment is awaiting moderation
2 MIN READ
0
Nutrient-dependent pheromone-controlled stickleback evolution
2 MIN READ
0
Functional coding variants are not mutations
2 MIN READ
0
Mirror neurons and microRNA: theory vs biological facts
4 MIN READ
0
Understanding the role of mutations and evolution
4 MIN READ
0
Alzheimer's, microRNAs, and olfaction
< 1 MIN READ
0
Evolution: innovations may have non-adaptive origins (sans mutations)
2 MIN READ
0
Pheromones and cancer
4 MIN READ
0
Are pheromones responsible for human body odour assessment?
2 MIN READ
0
Understanding evolution, adaptation, and phenotype
2 MIN READ
0
Nutrient-dependent / pheromone-controlled social structure
2 MIN READ
0
A flawed model: no tripartate synapse in the adult brain
3 MIN READ
0
Evolved development of olfactory systems
2 MIN READ
0
Epistasis as the primary factor in molecular evolution
4 MIN READ
0
Epigenetics and metabolism converge with pheromone production
2 MIN READ
0
Behavioral epigenetics in ecological context
2 MIN READ
0
Epigenetic maintenance of transcription / neuronal migration
2 MIN READ
0
Epigenetics and evolutionary success
< 1 MIN READ
0
Constraints on mutational drift
< 1 MIN READ
0
Epigenetic effects underlie sexual preferences IV
< 1 MIN READ
0
Epigenetic effects underlie sexual preferences III
3 MIN READ
0
Music to our ears and song in birds: a common sense approach
2 MIN READ
0
Thinking about what we know (or not)
2 MIN READ
0
A mathematical model of phenotypic cause, effects on genes, and affects on behavior
2 MIN READ
0
A newly discovered hypothalamic neurogenic niche in the brain
< 1 MIN READ
0
FDA: Just says NO — to ineffective drugs with side effects
< 1 MIN READ
0
Epigenetic effects underlie sexual preferences II
3 MIN READ
0
Is there a model for that?
3 MIN READ
0
One of the clearest examples of epigenetic effects
< 1 MIN READ
0
Pheromone-induced learning: a NEW model?
2 MIN READ
0
A Dictionary of Animal Behavior, Ecology and Evolution (excerpt)
2 MIN READ
0
Epigenetic effects underlie sexual preferences (duh, and food preferences)
2 MIN READ
0
Epigenetic effects on chromatin and regulatation of gene expression (in Archaea?)
2 MIN READ
0
The genetics myth meets the great pheromone myth
2 MIN READ
0
Systems biology met epigenetics two decades ago (not two days ago)
2 MIN READ
0
MicroRNA binds messenger RNA: Adaptive Evolution
< 1 MIN READ
0
Mutated genes and accidental intelligence?
< 1 MIN READ
0
Fatal attractions: a fear of pheromones
2 MIN READ
0
Genetic Influences on Epigenetically Effected Disease
2 MIN READ
0
Disorders of development: altered microRNA / messenger RNA balance
< 1 MIN READ
0
Eat like a monkey; act like a monkey
2 MIN READ
0
Human Pheromones: Epigenetic Effects on Hormones and Their Affects on Behavior
3 MIN READ
0
Forum discussions: the most complained about member (866 words)
3 MIN READ
0
Creation vs Evolution: Expression of a newly found gene
2 MIN READ
0
Epistasis: Epigenetic effects of nutrient chemicals and their metabolism to pheromones
< 1 MIN READ
0
Sex differences in Alzheimer's and everything else
2 MIN READ
0
The Knowing Nose: Chemical Signals Communicate Human Emotions
< 1 MIN READ
0
The logic of gene regulatory networks
3 MIN READ
0
Human Pheromones: Epigenetic Effects of Odors and Their Affects on Behavior
2 MIN READ
0
Creating new genes (and controlling diversity)
2 MIN READ
0
The end of random mutations theory
2 MIN READ
0
Epigenetic effects of viruses on cellular homeostasis
< 1 MIN READ
0
The same neural mechanisms are at work in worms and humans
2 MIN READ
0
Diet epigenetically effects development of the primate brain
< 1 MIN READ
0
Human pheromones determine our social behavior (REJECTED)
3 MIN READ
0
Human pheromones: epigenetic effects
< 1 MIN READ
0
Evolutionarily innovative DNA mistakes?
< 1 MIN READ
0
Pheromonemotionally speaking
2 MIN READ
0
Human Pheromones: theory vs models
3 MIN READ
0
Human pheromones: another attempt to change the concept
3 MIN READ
0
Human pheromones and the fusiform fooled you area for face recognition
3 MIN READ
0
The Missing Transgenerational Epigenetic Links
2 MIN READ
0
Playing with breasts (e.g, with a new THEORY of why only heterosexual men love them)
3 MIN READ
0
Society for Social Neuroscience Presentation scheduled 10/11/12
2 MIN READ
0
Epigenetic effects on stochastic gene expression and evolutionary psychology
2 MIN READ
0
The Ghost in Your Genes – BBC Horizon
< 1 MIN READ
0
Embracing galaxy-wide, cellular, and brain complexity
2 MIN READ
0
“Just-So” stories vs scientific explanations
2 MIN READ
0
Insect to human cognition
2 MIN READ
0
More complicated than 1-2-3
2 MIN READ
0
What to do with research results: Warning!
2 MIN READ
0
Mischaracterizations of adaptive evolution by psychologists and biologists
2 MIN READ
0
Human pheromones and the epigenetic effects of isolation
2 MIN READ
0
Transgenerational epigenetic inheritance of sexual orientation
2 MIN READ
0
Unconscious affects on incalculable genomic interactions
3 MIN READ
0
Natural selection and the behavior of whole organisms
< 1 MIN READ
0
Natural Selection and the Complexity of the Genome
2 MIN READ
0
Human pheromones: an epigenetic continuum from microbes to mammals
3 MIN READ
0
Biological facts constrain proponents of random mutations theory
2 MIN READ
0
Pheromones, Reciprocity, and Prosocial Behavior
3 MIN READ
0
Is the language of God the god problem? (1530 words)
6 MIN READ
0
The Epigenetics of Drug Development and Cholesterol Levels
< 1 MIN READ
0
Woefully ignorant politicians and popular science
2 MIN READ
0
Pheromones and multicellularity
< 1 MIN READ
0
Human pheromones and autism spectrum disorders (2)
2 MIN READ
0
Epigenetic alterations due to stress
< 1 MIN READ
0
Epigenetic effects on the evolution of behavior
2 MIN READ
0
Human pheromones and diet-mediated life-extension
< 1 MIN READ
0
CB Nemeroff: Off Restriction
< 1 MIN READ
0
Human pheromones and the definition of life
2 MIN READ
0
Human pheromones and autism spectrum disorders
2 MIN READ
0
Human pheromones, the social brain and simple math computations
2 MIN READ
0
Species specificity in gene expression in the brain
2 MIN READ
0
Children’s healthy diets lead to healthier IQ
< 1 MIN READ
0
Transgenerational epigenetic inheritance via nutrient chemicals
1 MIN READ
0
Human pheromones and IQ via neurogenic and socio-cognitive niche construction
2 MIN READ
0
Human pheromones, nutrition, DNA, epigenetics
2 MIN READ
0
Human pheromones are like sugar (and spice)
< 1 MIN READ
0
Niche construction: ecological, social, neurogenic, and socio-cognitive
2 MIN READ
0
Oxytocin for everything?
2 MIN READ
1
Human Pheromones and Free Will: a response to Gazzaniga
2 MIN READ
0
Human pheromones and multi-level selection
< 1 MIN READ
0
Where is the love?
3 MIN READ
0
Human pheromones and the viruses that make us
2 MIN READ
0
A detailed model of physical reality and natural selection (722 words)
3 MIN READ
0
Evolution of the nervous system: conserved GnRH
< 1 MIN READ
0
Is modeling natural selection as a sensing agent reasonable?
3 MIN READ
0
Human Pheromones and Comparative Epigenomics
2 MIN READ
0
Aging Is Recorded in Our Genes
2 MIN READ
0
Human Pheromone Facts and the Out of Africa Theory
3 MIN READ
0
Smelling the difference between young and old
< 1 MIN READ
0
Human pheromones and the visual appeal of other people (Part Four)
3 MIN READ
0
A thought experiment
3 MIN READ
0
Human pheromones: Olfaction, odor receptors, learning, memory, mood, brains and behavior
< 1 MIN READ
0
A nutrient chemical and pheromone-dependent neurogenic niche
2 MIN READ
0
Human pheromone- and nutrient-dependent brain development and behavior
2 MIN READ
0
Human Pheromones: The smell of the scientific universe expanding
< 1 MIN READ
0
More than speculation: adaptive evolution in species from microbes to man
2 MIN READ
0
Adam’s rib: Pheromones and the adaptive evolution of human sexuality
2 MIN READ
0
Human pheromones and your immune system
< 1 MIN READ
0
Human Pheromones in Socioaffective Neuroscience & Psychology
2 MIN READ
0
Pheromones and the “Bruce effect” in wild or domesticated horses and primates
2 MIN READ
0
Love is a receptor-mediated event: the pervasive influence of the late Robert L. Moss
3 MIN READ
0
They are what they eat, and so are we
2 MIN READ
0
Exercise Alters Epigenetics
2 MIN READ
0
How memory works
2 MIN READ
0
A Preoccupation With Modesty…
2 MIN READ
0
Creation of different species without physical barriers (Part Two)
2 MIN READ
0
Pheromones: from microbes to man
< 1 MIN READ
0
The biology of behavior: Seeing more clearly
< 1 MIN READ
0
Researchers Investigate Epigenetic Patterns In Multiple Conditions
< 1 MIN READ
0
Genes not culture drive humans forward (an antithetical approach)
< 1 MIN READ
0
Effects of pheromones on menstrual synchrony and a gay man
2 MIN READ
0
Did viruses make mammals communicate with pheromones?
2 MIN READ
0
Do Pheromones Play a Role in Our Sex Lives: Scientific American
< 1 MIN READ
0
Human Pheromones: 2010 Powerpoint slides and 2011 poster presentation
2 MIN READ
0
Epigentics and the baby connectome
2 MIN READ
0
Cookbook chemistry: food odors and hormones
< 1 MIN READ
0
Epigenetics and the connectome
2 MIN READ
0
Human pheromones and food odors
< 1 MIN READ
0
Pheromones and the Evolution of Drug Resistance
< 1 MIN READ
0
Human pheromones provide clues to personality
< 1 MIN READ
0
Show me the science!
5 MIN READ
0
How pheromones work (in 57 words or more)
< 1 MIN READ
0
Pheromones and falling in love (circa 1974)
< 1 MIN READ
0
Pheromones, LH, neurogenesis, learning, memory, behavior
< 1 MIN READ
0
Pheromone-enhanced behavior in sexually inexperienced females
2 MIN READ
0
Epigenetic ‘memory’ key to nature versus nurture
< 1 MIN READ
0
Avian ‘Axe effect’ attracts attention of females and males
2 MIN READ
0
Study results: Human pheromones affect women’s behavior
< 1 MIN READ
0
Human pheromones: social odors and food odors
< 1 MIN READ
0
Honeybees, food odors, and perfume
< 1 MIN READ
0
Avian recognition of human faces
< 1 MIN READ
0
Human Pheromones Affect Women's Behavior
2 MIN READ
0
A lesser role for olfaction and pheromones in humans?
1 MIN READ
0
Creating a Myth
< 1 MIN READ
0
Study results: Human pheromones influence human behavior
2 MIN READ
0
Pheromones in birds, other mammals, and us
< 1 MIN READ
0
The great pheromone myth revisited (including biological factors in the development of sexuality)
4 MIN READ
0
The great pheromone myth revisited (including biological factors in the development of sexuality)
4 MIN READ
0
Social selection: genetic contribution of viruses to life on earth
< 1 MIN READ
0
Wikipedia comments by James V. Kohl
5 MIN READ
0
Excellent new book about pheromones
< 1 MIN READ
0
Study design| Psychophysics experiment | Perspective on conditioning
< 1 MIN READ
0
Adrenarche and odor preferences
2 MIN READ
0
For ethologists: Observation does not trump molecular biology
2 MIN READ
0
Human Pheromones and The Philosophy of Science
2 MIN READ
0
Genes, Hormones, and Behavior (for other scientists)
4 MIN READ
0
Love at first sight: Selective breeding, pheromones, and Divine intervention
3 MIN READ
0
A fundamental attribution error: Love at first sight
3 MIN READ
0
Human Pheromones
< 1 MIN READ
0
Pheromones and olfaction in the birds and the bees: from yeasts to you
2 MIN READ
0
Debunking the myth of pheromones
< 1 MIN READ
0
Tears and fears of pheromones
2 MIN READ
0
Exotic Pheromones and Concentrates
2 MIN READ
0
Marketing vs. the truth about human pheromones
2 MIN READ
0
Epigenetics, science fiction, and scientific fact
2 MIN READ
0
Results: increased flirtatious behavior and self-reported attraction
2 MIN READ
0
Is it what he or she sees in you, or is it your pheromones?
2 MIN READ
0
Product comparisons and human pheromone sciences
3 MIN READ
0
Conditioning of the human pair bond by human pheromones
< 1 MIN READ
0
Conditioned sexual arousal to odor
2 MIN READ
0
Scent signals
< 1 MIN READ
0
Pheromones and signature mixtures
2 MIN READ
0
Comparative biology of pheromonal communication
< 1 MIN READ
1
Food preferences and mate preferences
< 1 MIN READ
0
Presentation on Human Pheromones delivered on August 3, 2010
2 MIN READ
0
Sold on the human VNO?
< 1 MIN READ
0
Effects of pheromones attributed to touch (in mice)
< 1 MIN READ
0
How pheromones work by James V. Kohl
2 MIN READ
0
Pheromones of pregnant women effect prolactin in men
< 1 MIN READ
0
Another book author's comment on "The Great Pheromone Myth"
< 1 MIN READ
0
An attempt to change the concept
< 1 MIN READ
0
Luteinizing hormone, sex, and smell (18 years later)
2 MIN READ
0



[…] Epigenetic regulation of aging by glycine and GnRH […]